DREAMING IS A COMPLEX AND MYSTERIOUS ASPECT OF human consciousness, with dreams offering a window into our unconscious mind. While the exact functions of dreams remain elusive, they are considered essential for emotional processing, memory consolidation and even problem-solving. However, when it comes to traumatic brain injury (TBI), the effects on dreaming are not well understood. TBI, which occurs due to an external force to the head, can cause a range of cognitive, emotional and physical impairments. It can also have a profound impact on the dreaming process, which is intricately tied to the brain’s ability to function. This article explores how TBI affects dreaming, the changes in dream patterns and the potential reasons behind these alterations.
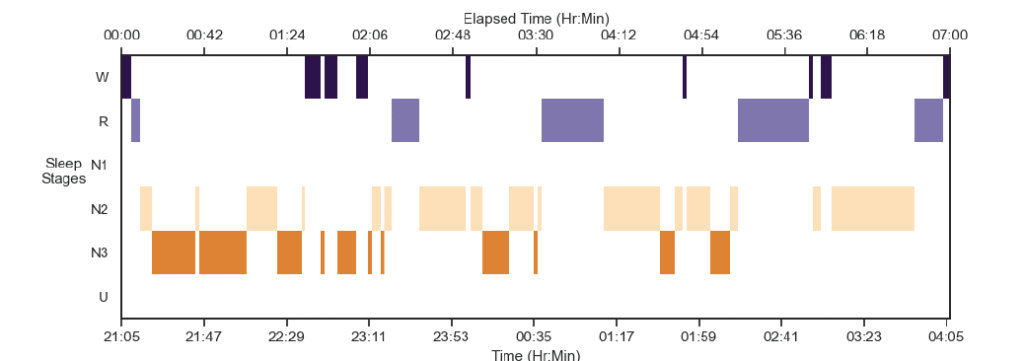
Sleep stages graph (hypnogram) above shows the different stages of sleep of participants during their session.
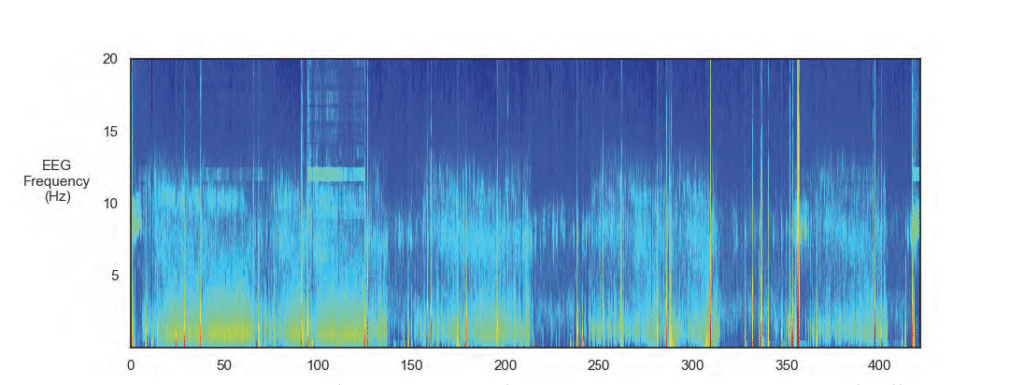
The EEG spectogram image above shows the intensity of brainwave activity over time. High-intensity regions are indicated by tones of red, yellow and green, and low-intensity regions are indicated by tones of blue.
UNDERSTANDING DREAMING AND THE BRAIN
Dreams are primarily associated with rapid eye movement (REM) sleep, a stage of sleep that is crucial for restorative processes in the brain. During REM sleep, brain activity is high, and the brain undergoes various functions, including the processing and consolidation of memories. It is during REM sleep that vivid and often surreal dreams occur. Non-REM sleep stages, especially deep sleep, also play a role in memory consolidation, physical restoration and the maintenance of cognitive functions.
The brain structures that play an essential role in dreaming include the prefrontal cortex, the limbic system (which controls emotions) and the hippocampus (which is involved in memory formation). These regions work together to create the vivid scenarios that we experience while dreaming. Traumatic brain injury can disrupt these brain structures, resulting in changes to the dreaming process, affecting both the content and quality of dreams.

THE IMPACT OF TBI ON DREAMING
The effect of TBI on dreaming varies depending on the severity of the injury, the regions of the brain affected and whether the individual is recovering from the injury. However, several common trends have emerged in research that help to explain the link between TBI and changes in dreaming.
Disruption in REM Sleep
One of the primary effects of TBI on dreaming is a disruption of the sleep cycle, especially REM sleep. Many individuals with TBI report experiencing difficulty entering or maintaining REM sleep, which in turn affects the intensity and frequency of dreams. Some individuals with TBI may experience less REM sleep overall, which reduces the opportunity for dreaming. On the other hand, some individuals report fragmented or disrupted REM sleep, leading to sleep disturbances that may cause nightmares or vivid dreams.
The disturbance in REM sleep following TBI is often linked to the brain’s inability to regulate sleep cycles effectively. Damage to brain regions such as the hypothalamus, which controls the circadian rhythm, and the brainstem, which regulates REM sleep, can result in sleep fragmentation and other disturbances that affect dreaming.

Changes in Dream Content
The content of dreams can be profoundly altered after a traumatic brain injury. Dreams may become more bizarre, fragmented or distressing. Some individuals with TBI report an increase in violent or frightening dreams, including nightmares, possibly due to heightened emotional responses to stress or trauma experienced during the injury. The emotional part of the brain, particularly the amygdala, may be affected by TBI, which could make emotions such as fear, anxiety and anger more dominant in dreams.
Moreover, people with TBI may experience more frequent dreams that revolve around the injury itself or the events surrounding it. These types of dreams may be related to post-traumatic stress, a common condition following a TBI. Dreams may replay the traumatic incident or present variations of the injury experience, leading to increased distress and anxiety. Such dreams are often reflective of the brain’s attempt to process and integrate traumatic memories.
Sleep Apnea and Other Sleep Disorders
Sleep disorders, including sleep apnea, are common in individuals with TBI. Sleep apnea is a condition in which a person’s breathing repeatedly stops and starts during sleep. This leads to reduced oxygen levels in the blood, further disrupting the sleep cycle and negatively affecting REM sleep. The lack of quality REM sleep can significantly impact dreaming, causing a reduction in dream intensity or altering the nature of dreams.
TBI can contribute to the development of sleep apnea by affecting the brainstem, which plays a role in regulating breathing. In addition to sleep apnea, other sleep disorders such as insomnia and restless leg syndrome may also develop after TBI, further affecting sleep quality and, by extension, the dreaming process.
NEUROCHEMICAL CHANGES AND THEIR EFFECT ON DREAMS
The brain’s neurotransmitter systems play a crucial role in regulating sleep and dreaming. TBI can result in alterations in the levels of neurotransmitters such as serotonin, dopamine and acetylcholine, which are all involved in the regulation of sleep and dream activity. Damage to these systems may lead to abnormal sleep cycles, less REM sleep and changes in the emotional tone of dreams.
Serotonin, for example, is critical for maintaining healthy sleep patterns and mood regulation. TBI-related disruptions in serotonin levels may contribute to sleep disturbances, nightmares and emotional instability in dreams. Similarly, changes in dopamine levels can affect motivation, reward systems and mood regulation, leading to more vivid or unusual dreams. Alterations in acetylcholine, a neurotransmitter essential for REM sleep, can directly impact the quality and frequency of dreaming.

NEUROPLASTICITY AND DREAMING POST-TBI
Neuroplasticity, the brain’s ability to reorganize itself and form new neural connections, plays a role in recovery after a traumatic brain injury. As the brain heals and compensates for areas of damage, it may restore some functions, including the ability to engage in normal sleep cycles and experience regular dreams. However, the extent of this recovery depends on the severity of the injury, the areas of the brain affected and the individual’s age and overall health.
In some cases, as neuroplasticity allows the brain to compensate for damage, individuals with TBI may experience a return of more vivid or coherent dreams. Conversely, some individuals may experience persistent disruptions in dreaming as the brain continues to adapt to the injury. The process of neuroplasticity is unique to each individual, and its impact on dreaming remains an area of ongoing research.
POTENTIAL TREATMENTS FOR DREAMING DISTURBANCES FOLLOWING TBI
While there is no definitive treatment to restore normal dreaming after a traumatic brain injury, several strategies may help manage the associated sleep disturbances and improve sleep quality, which, in turn, can support healthier dreaming.
Cognitive Behavioral Therapy for Insomnia (CBT-I): This type of therapy is often used to treat sleep disturbances in individuals with TBI. CBT-I helps individuals identify and address thoughts and behaviors that interfere with sleep, leading to improvements in overall sleep quality, which may also have a positive effect on dreaming.
Pharmacological Interventions: Medications such as antidepressants, antianxiety drugs or sleep aids may
be prescribed to individuals with TBI who suffer from nightmares or other sleep disturbances. These medications may help to regulate sleep patterns and reduce the frequency of nightmares.
Relaxation Techniques: Techniques such as meditation, deep breathing and progressive muscle relaxation may help individuals with TBI reduce stress and anxiety before sleep, leading to improved sleep quality and potentially less disturbing dreams.
Sleep Hygiene Education: Establishing a consistent sleep routine and creating a calm and comfortable sleep environment can improve sleep quality, leading to better recovery and less disruption to dreaming. Sleep hygiene strategies may include maintaining a consistent sleep-wake cycle, avoiding caffeine and alcohol before bedtime and minimizing screen time in the hours leading up to sleep.
Nightmare Management: Individuals who suffer from frequent nightmares related to TBI may benefit from therapies that focus on trauma resolution, such as EMDR (eye movement desensitization and reprocessing) or trauma-focused cognitive-behavioral therapy. These approaches aim to reduce the emotional impact of traumatic memories, potentially decreasing the frequency and intensity of nightmares.
IN CONCLUSION
The effects of traumatic brain injury on dreaming are varied and complex, reflecting the intricate nature of the brain’s sleep and memory systems. TBI can lead to disruptions in REM sleep, changes in dream content, increased nightmares and memory difficulties related to dream recall. These disturbances are influenced by various factors, including neurochemical changes, emotional responses to trauma and disruptions in brain regions responsible for sleep regulation.
Although many individuals with TBI experience significant changes to their dreams, there are effective strategies for managing these disturbances, including cognitive behavioral therapies, pharmacological interventions and lifestyle changes. By addressing the underlying sleep issues, it is possible to improve sleep quality, reduce nightmares and enhance the overall healing process following a traumatic brain injury. As research into the relationship between TBI and dreaming continues, a better understanding of these processes may lead to more targeted treatments and interventions.
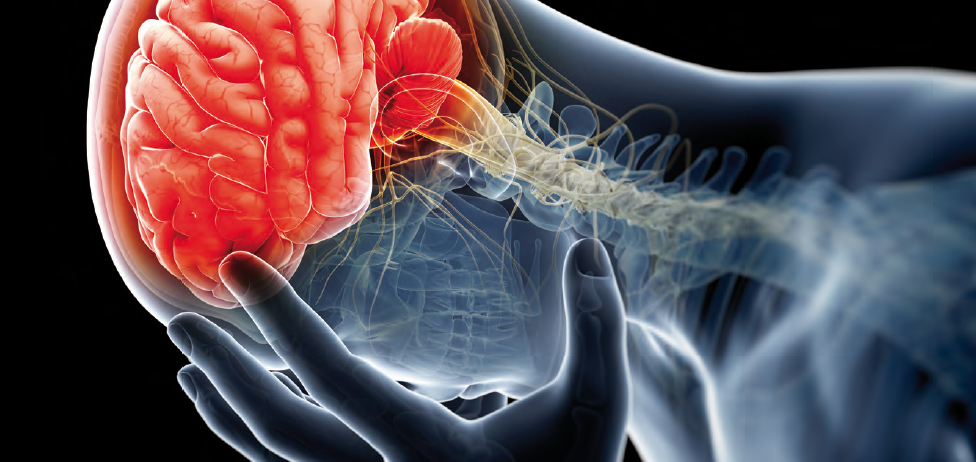
Traumatic brain injury (TBI) is a serious and often life-threatening condition that results from a sudden blow or jolt to the head, causing damage to the brain. While many individuals who experience a TBI recover with appropriate treatment, in some cases, the injury can lead to death. The severity of the brain injury and the specific areas of the brain that are affected play a significant role in determining the outcome. Understanding how and why TBI can lead to death requires an exploration of the mechanisms of injury, the types of TBIs and the complications that can arise in the aftermath of a traumatic event.
MECHANISMS OF TRAUMATIC BRAIN INJURY
The brain is a delicate organ, and any significant trauma to the head can cause a range of injuries. TBI can occur through several mechanisms, including direct impact, acceleration-deceleration forces and rotational forces. A direct impact, such as a blow to the head from a fall or a car accident, can cause localized damage to the brain. The brain is cushioned by cerebrospinal fluid within the skull, but when an external force is strong enough, the brain can collide with the inside of the skull, causing bruising (contusion), swelling and even bleeding (hemorrhage).
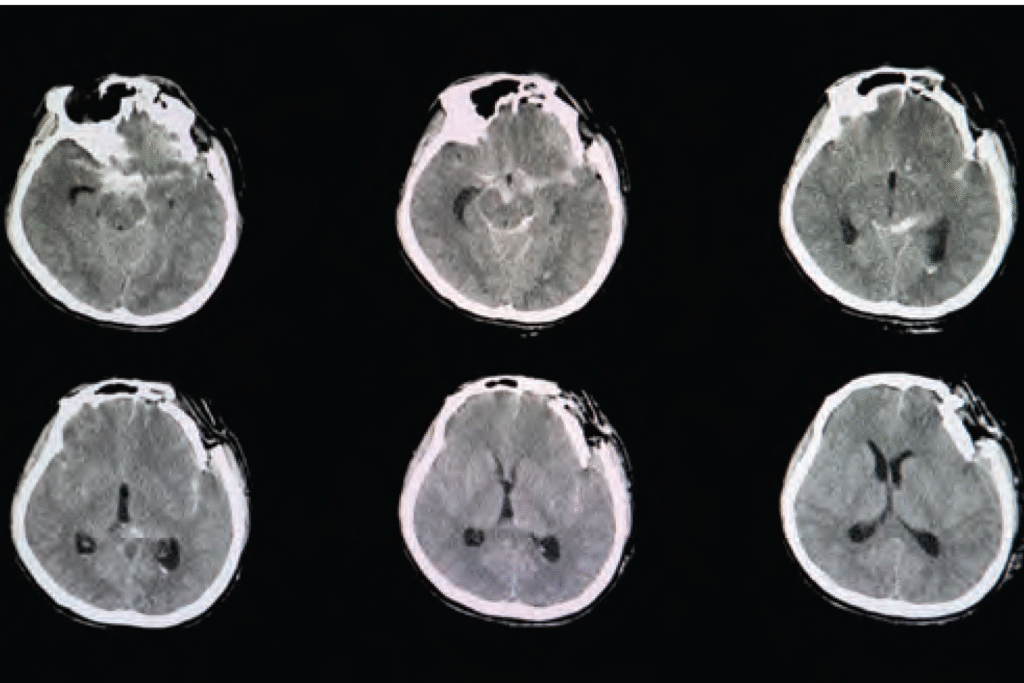
CT brain scan image of a patient with severe skull depression fracture at left frontal parietal area with left zygomatic arch and both maxillary sinuses fractures with small subarachnoidal hemorrhage.
Acceleration-deceleration injuries occur when the head is rapidly moved back and forth, as in a car crash or sports-related injury. This rapid motion causes the brain to shift within the skull, leading to damage to brain tissue, blood vessels and nerve fibers. Rotational forces, such as those caused by a sudden twisting motion, can tear nerve fibers (axons), resulting in diffuse axonal injury (DAI). DAI is one of the most severe forms of TBI and often results in long-term impairments or death. These mechanisms can cause damage to different regions of the brain, each of which plays a crucial role in basic life functions such as breathing, heart rate and consciousness.
TYPES OF TRAUMATIC BRAIN INJURY
TBI is classified into three main categories based on severity: mild, moderate and severe. Mild TBI, often referred to as a concussion, typically involves a brief loss of consciousness or confusion, with recovery expected within days to weeks. Moderate and severe TBIs, however, carry a much higher risk of complications and death. Moderate TBI can lead to lasting cognitive deficits and physical impairments, while severe TBI often involves significant brain swelling, bleeding and widespread tissue damage that can be fatal.
One of the most dangerous forms of TBI is a penetrating injury, in which an object, such as a bullet or sharp object, enters the skull and damages brain tissue. These injuries are often fatal because they cause immediate, severe damage to critical brain structures, leading to uncontrollable bleeding, infection and loss of function in vital areas of the brain. Subdural and epidural hematomas, which involve the accumulation of blood between the brain and the skull, are also life-threatening conditions that can result from moderate to severe TBI. These blood clots can put pressure on the brain, reducing blood flow and oxygen supply to vital brain regions, leading to brain death.
Brain Swelling and Increased Intracranial Pressure. One of the primary reasons that TBI can lead to death is brain swelling (cerebral edema), which can increase intracranial pressure (ICP). When the brain swells due to injury, there is limited space within the rigid skull to accommodate the expanding tissue. As a result, the pressure inside the skull rises, which can restrict blood flow to the brain and decrease the supply of oxygen and nutrients. This can lead to widespread brain cell death and irreversible brain damage.
COMPLICATIONS LEADING TO DEATH
In addition to brain swelling and increased ICP, there are several other complications that can arise from TBI and lead to death. These complications include hemorrhaging (bleeding) within the brain, infections and seizures. Intracranial bleeding, such as subdural or epidural hematomas, can quickly worsen and lead to irreversible brain damage. Hemorrhages can occur immediately after the injury or develop over time, making it crucial for medical professionals to monitor individuals with severe TBI closely.
Infections, particularly in cases of open head wounds or penetrating injuries, can also be fatal. Infections can spread rapidly within the brain, causing inflammation, tissue destruction and sepsis, which can lead to multi-organ failure and death. Seizures are another common complication of TBI, especially in individuals with moderate to severe injuries. Seizures can further damage brain tissue, increase ICP and contribute to other life-threatening conditions.
Traumatic brain injury can cause death through a combination of direct damage to the brain, swelling, bleeding and secondary complications such as infection and seizures. The severity of the injury, the areas of the brain affected and the individual’s overall health determine the likelihood of survival and recovery. Severe TBIs, particularly those involving significant brain swelling, hemorrhaging or penetration, carry the highest risk of death. Early intervention, medical management to control swelling and bleeding, and surgery to address complications can improve survival rates, but TBI remains a leading cause of death and long-term disability worldwide. Understanding the mechanisms of TBI and the risks involved highlights the importance of prevention, early detection and prompt medical treatment to reduce the potentially fatal consequences of head injuries.
Awake craniotomy is an advanced surgical technique that is increasingly used to treat traumatic brain aneurysms, which are abnormal bulges or weakened areas in the walls of blood vessels within the brain and caused by traumatic injury. These aneurysms are particularly dangerous because they can rupture, leading to severe brain hemorrhage which may cause irreversible damage or even death. Traditionally, treating such aneurysms required traditional craniotomy, in which the skull is opened to access the brain, but awake craniotomy offers a more precise and safe alternative, especially when the aneurysm is located near critical brain structures.
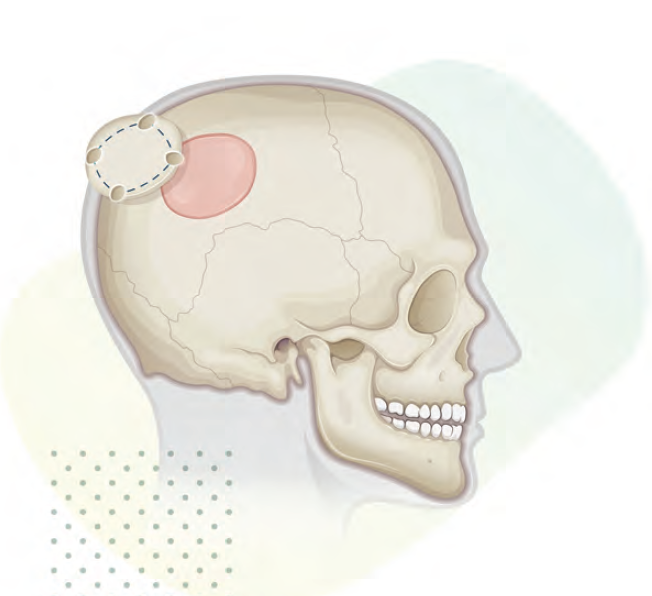
A craniotomy is a surgical operation in which a bone flap is temporarily removed from the skull to access the brain.
During an awake craniotomy, the patient is kept conscious throughout the procedure, but local anesthesia is administered to numb the scalp and skull. The patient is sedated enough to remain calm and comfortable, but remains alert and responsive for most of the surgery. This conscious state allows the surgeon to interact directly with the patient, performing neurological tests during the procedure. If the aneurysm is near regions of the brain responsible for essential functions such as speech, movement, or cognition, the surgeon can monitor the patient’s responses to stimulation, ensuring that vital brain areas are not damaged during the surgery.
For patients with traumatic aneurysms, awake craniotomy allows the surgical team to more effectively target the aneurysm while avoiding harm to crucial brain functions. For example, during surgery, the surgeon may ask the patient to speak or move a limb while stimulating specific brain areas to assess which parts of the brain control those functions. This real-time feedback enables the surgical team to navigate sensitive areas more carefully. The surgeon can make adjustments if they notice any adverse effects or if the patient shows signs of discomfort. This level of precision is particularly important for aneurysms located in complex or deep regions of the brain, where traditional surgery might risk significant damage to nearby structures.
One of the significant advantages of awake craniotomy is the ability to monitor the patient’s neurological status during surgery. In traditional surgeries, patients are typically under general anesthesia, making it impossible for the surgical team to assess the patient’s cognitive and motor functions in real time. In contrast, with awake craniotomy, the surgeon can adjust the procedure dynamically, reducing the risk
of complications such as paralysis, speech difficulties, or cognitive impairment. By ensuring that the brain is undamaged, the technique can result in better long-term outcomes and fewer complications after surgery. Patients are often able to recover more quickly and with less residual neurological damage, making this method particularly beneficial for those with traumatic brain injuries.
While awake craniotomy has many advantages, it also comes with challenges. The patient must be able to cooperate throughout the procedure, which may be difficult for some individuals, particularly those who experience anxiety or discomfort. Additionally, the procedure requires highly skilled surgical teams who can effectively communicate with the patient and monitor the brain’s response to stimulation. Despite these challenges, awake craniotomy remains a groundbreaking technique that significantly improves the treatment of traumatic aneurysms, offering a safer, more effective alternative to traditional surgical methods. By allowing surgeons to operate with increased precision and real-time feedback, awake craniotomy has become a key tool in treating brain aneurysms while minimizing the risk of long-term complications.
Stroke is a leading cause of disability and death worldwide. It occurs when there is a sudden interruption of blood flow to the brain, resulting in the death or damage of brain cells. While strokes have long been categorized as vascular events, they share similarities with traumatic brain injuries (TBIs) in their effects on brain tissue. Specifically, a stroke is now recognized as a type of non-traumatic brain injury (nTBI), a category that is not caused by direct physical trauma to the brain. This article explores the neuroscience behind stroke, its impact on the brain and why it is classified as an nTBI.
WHAT IS A STROKE?
A stroke occurs when there is a disruption in the blood supply to a part of the brain. This disruption can be due to two main mechanisms:
Ischemic Stroke: This type of stroke is caused by a blockage or narrowing of the blood vessels that supply the brain. A clot can form either in the brain itself (thrombotic stroke) or travel from another part of the body, such as the heart (embolic stroke).
Hemorrhagic Stroke: This occurs when a blood vessel in the brain bursts, leading to bleeding within the brain. The pooling of blood puts pressure on surrounding brain tissues, causing damage.
Both types of strokes share a common outcome: a lack of oxygen and nutrients to brain tissue, leading to cell death and neurological dysfunction. The primary difference between the two types is the mechanism causing the disruption in blood flow.
Ischemia and Oxygen Deprivation
When blood flow to a part of the brain is interrupted, the affected tissue becomes deprived of oxygen and glucose. These are essential for neurons to produce energy and function normally. Within minutes of ischemia, brain cells begin to suffer damage. The immediate effect is a phenomenon called cerebral ischemia, which causes a range of toxic processes within the brain.
THE NEUROSCIENCE OF STROKE: HOW IT AFFECTS THE BRAIN
To understand why stroke is considered an nTBI, it is important to examine the physiological mechanisms that occur during a stroke. Regardless of the type of stroke, the fundamental problem is the disruption of cerebral blood flow, which triggers a cascade of cellular and molecular events in the brain. Hemorrhagic Stroke: This occurs when a blood vessel in the brain bursts, leading to bleeding within the brain. The pooling of blood puts pressure on surrounding brain tissues, causing damage.
Both types of strokes share a common outcome: a lack of oxygen and nutrients to brain tissue, leading to cell death and neurological dysfunction. The primary difference between the two types is the mechanism causing the disruption in blood flow.
At the cellular level, neurons begin to experience energy failure, primarily due to a lack of adenosine triphosphate (ATP), which is crucial for maintaining normal cellular functions. Without ATP, the cell’s ion pumps malfunction, causing an imbalance of ions across the cell membrane. This leads to a phenomenon called excitotoxicity, where an excessive release of neurotransmitters, especially glutamate, results in an influx of calcium ions into the neuron. High calcium levels activate various enzymes that break down cellular structures, leading to cell death.
Stroke leads to inflammation in the affected brain region. This inflammation, called neuroinflammation, is triggered by the damage to brain cells and blood vessels. The activation of microglia, the brain’s resident immune cells, is a key component of this inflammatory response. Initially, microglia attempt to clear up dead cells and debris, but if the inflammation is prolonged, it can contribute to further brain injury by releasing pro-inflammatory cytokines and other toxic molecules.
Neuroinflammation is not only harmful in the acute phase of stroke but also contributes to long-term brain damage and recovery. The chronic inflammation that follows stroke has been implicated in the development of neurological deficits, cognitive decline and the progression of neurodegenerative diseases.
STROKE AS A NON-TRAUMATIC BRAIN INJURY (nTBI)
Traditionally, TBIs have been associated with external physical forces such as blows to the head, falls, or accidents. These external
forces result in direct trauma to the brain, leading to tissue damage and dysfunction. However, stroke, while not caused by a direct mechanical impact, can produce similar effects on brain tissue and functions. This has led to the recognition of stroke as a non-traumatic brain injury (nTBI).
An nTBI refers to any type of brain injury that is not the result of an external physical force but still leads to structural or functional damage to the brain. Strokes, whether ischemic or hemorrhagic, fit this definition because they result in significant brain injury due to the interruption of blood flow without any direct external impact on the brain.
WHY STROKE IS CONSIDERED AN NTBI
There are several reasons why stroke is categorized as an nTBI:
Similar Pathophysiology: Like traumatic brain injuries, stroke leads to neuronal cell death, neuroinflammation and long-term changes in brain structure and function. Both conditions cause damage to neurons and glial cells, leading to neurological deficits, cognitive impairments and emotional disturbances.
No External Trauma: Unlike TBIs, which are caused by external physical forces, strokes occur due to internal processes, such as blood clots or ruptured blood vessels. There is no direct impact on the skull or brain tissue from an external object, yet the effects on brain function can be just as devastating as a TBI.
Long-Term Consequences: Both stroke and TBI often result in long-term functional impairments, including motor deficits, sensory disturbances, cognitive decline and mood disorders. The recovery process for both conditions can be slow, requiring extensive rehabilitation and often leading to permanent disability.
Overlap in Risk Factors: Many of the risk factors for stroke overlap with those of TBI. For example, age, hypertension, diabetes and cardiovascular diseases are risk factors for both strokes and TBIs. Furthermore, individuals who have suffered a TBI are at an increased risk for stroke, further emphasizing the connection between the two conditions.
Impact on Brain Networks: Both stroke and TBI can disrupt the functioning of large-scale brain networks, affecting motor, sensory and cognitive functions. These disruptions can lead to widespread changes in brain connectivity, which is often associated with neurological deficits and difficulties in recovery.
CLINICAL IMPLICATIONS AND TREATMENT OF STROKE AS AN NTBI
Recognizing stroke as an nTBI has important clinical implications. It highlights the need for timely intervention to prevent further brain damage and improve outcomes. For example, the use of thrombolytic therapy (clot-busting drugs) and mechanical thrombectomy in ischemic stroke can restore blood flow and limit the extent of brain damage, much like how early intervention in TBI can prevent secondary injury.
Additionally, the rehabilitation of stroke patients often involves similar strategies used for TBI patients, including physical therapy, occupational therapy and neuropsychological interventions. The goal is to help patients recover lost functions and adapt to any remaining disabilities.
IN CONCLUSION
Stroke is a complex neurological event that results in the disruption of blood flow to the brain, leading to cell death, inflammation, and long-term neurological impairment. While strokes are caused by vascular issues and not external trauma, they share many similarities with traumatic brain injuries in terms of
their pathophysiology, clinical outcomes and impact on brain function. For these reasons, stroke is increasingly recognized as a form of non-traumatic brain injury (nTBI), and understanding its mechanisms can help improve treatment and rehabilitation strategies for those affected. By viewing stroke through the lens of nTBI, we can better address the needs of patients, leading to improved care and recovery outcomes.
Mild traumatic brain injury (mTBI), often referred to as a concussion, is one of the most common and underdiagnosed injuries in both sports and everyday life. Traditionally, concussions have been diagnosed based on clinical symptoms and patient history, which may often be subjective and prone to misinterpretation. This uncertainty has prompted extensive research to find more objective and accurate diagnostic tools for mTBI, with molecular markers emerging as a promising solution. The identification of these biomarkers could potentially revolutionize the way concussions are diagnosed, offering a path toward definitive, objective diagnosis. This article explores the current state of research on molecular markers for mTBI and their role in making concussion diagnosis a reality.
THE CHALLENGE OF DIAGNOSING MTBI
A concussion results from a blow to the head or a jolt to the body that causes the brain to move rapidly inside the skull, leading to temporary disruption in brain function. The symptoms of a concussion are wide-ranging, including headaches, dizziness, confusion, nausea and cognitive difficulties such as memory problems and difficulty concentrating. Importantly, many symptoms are not visible, and they can evolve over hours or days, making it challenging to assess the extent of the injury in real time.
Currently, the diagnosis of mTBI relies heavily on clinical evaluations, such as the Glasgow Coma Scale (GCS), symptom checklists, and imaging tests like CT or MRI scans. However, these methods have limitations. GCS scores are useful in assessing the severity of brain injury, but they do not differentiate between mild and moderate brain injuries effectively. Imaging techniques, while excellent at detecting more severe brain injuries, often fail to show any abnormalities in the case of mTBI, as the damage caused by concussions is typically microscopic.
This lack of diagnostic precision can lead to underdiagnosis, where individuals with concussions continue to engage in physical activity, risking further injury, or overdiagnosis, where individuals are sidelined unnecessarily. Both scenarios highlight the need for a more objective method to diagnose concussions accurately.
THE PROMISE OF MOLECULAR MARKERS
In recent years, research into the molecular mechanisms underlying mTBI has revealed that certain proteins and molecules are released into the bloodstream or cerebrospinal fluid (CSF) shortly after a brain injury. These biomarkers, or molecular markers, can provide crucial information about the extent and nature of brain injury. Unlike traditional methods, molecular markers offer a window into the biological changes occurring at the cellular level following a concussion.
The use of molecular markers as a diagnostic tool for mTBI holds immense promise because it can provide a more definitive and objective measure of injury. Unlike symptom-based assessments, which may be influenced by the patient’s ability to report symptoms and the subjective interpretation of healthcare providers, molecular markers are tangible and measurable indicators of physiological changes. Additionally, the detection of molecular markers can help monitor recovery, guide clinical decisions and aid in preventing second-impact syndrome, a potentially fatal condition that occurs when an individual sustains a second concussion before the first one has healed.
KEY MOLECULAR MARKERS FOR MTBI
Numerous molecular markers have been identified in the search for reliable diagnostic tools for mTBI. These markers typically fall into categories based on the nature of the substances they represent, such as neuronal proteins, glial markers, or molecules associated with inflammation. Below are some of the most promising molecular markers:
- Glial Fibrillary Acidic Protein (GFAP)
GFAP is a protein found in the glial cells of the brain, which provide support and nourishment to neurons. When the brain is injured, GFAP is released into the bloodstream as glial cells are damaged. Elevated levels of GFAP have been shown to correlate with the severity of brain injury. In fact, research has shown that GFAP levels can be measured in the blood within hours
of a concussion and provide valuable insights into the injury’s severity. GFAP, along with other markers, may become a key biomarker for mTBI diagnosis. - S100B Protein
S100B is a calcium-binding protein that is primarily expressed in glial cells and neurons. When the brain is injured, S100B is released into the bloodstream, making it a potential marker for detecting mTBI. Elevated levels of S100B in the blood have been associated with both mild and severe brain injuries. This biomarker has been widely studied, and although its specificity for mTBI remains a topic of ongoing research, its use in conjunction with other markers could significantly improve diagnostic accuracy. - Ubiquitin C-terminal Hydrolase L1 (UCH-L1)
UCH-L1 is an enzyme found in neurons that plays a role in protein degradation. In the event of neuronal injury, UCH-L1 is released into the bloodstream. Several studies have demonstrated that increased levels of UCH-L1 in blood samples are strongly associated with brain injury, and elevated concentrations have been observed following both acute and subacute phases of mTBI. UCH-L1 is particularly promising because it can be detected shortly after the injury and may provide an early indicator of concussion severity. - Tau Protein
Tau is a protein that stabilizes microtubules in neurons. Following a brain injury, tau can become hyperphosphorylated, leading to its detachment from microtubules and subsequent release into the bloodstream. Elevated tau levels are typically associated with more severe forms of brain injury and are considered a hallmark of neurodegenerative diseases such as Alzheimer’s. However, its role in mTBI has garnered significant attention. Elevated tau levels can serve as a potential marker for detecting concussion-related brain injury, particularly when combined with other biomarkers.
- Neurofilament Light Chain (NfL)
NfL is a structural protein found in neurons, and its release into the bloodstream has been linked to axonal injury. As axons are damaged following mTBI, NfL levels in the blood rise. Several studies have demonstrated that NfL concentrations are elevated in patients with mTBI and can provide valuable information about the extent of axonal injury. NfL may offer a broad and reliable method for detecting brain injuries, especially mild ones, that would not otherwise be visible using conventional imaging techniques.
CLINICAL IMPLICATIONS OF MOLECULAR MARKERS FOR MTBI DIAGNOSIS
The ability to identify and measure these molecular markers in blood or CSF opens up several exciting possibilities for improving concussion diagnosis and management. One of the most significant advantages of molecular markers is their ability to detect mTBI early, even in the absence of visible symptoms or abnormalities on standard imaging. This early detection could allow healthcare professionals to make timely decisions about whether an athlete or patient should rest, undergo further testing, or return to activity.
Furthermore, molecular markers can be used to track recovery. By measuring biomarker levels over time, clinicians can assess whether the brain is healing appropriately or if additional treatment or rest is needed. This monitoring could help prevent the long-term consequences of multiple concussions, including chronic traumatic encephalopathy (CTE) and other neurodegenerative diseases.
Molecular markers may also reduce the reliance on subjective symptom reporting, which may be inconsistent, particularly among athletes who may be reluctant to report their symptoms due to the desire to continue playing. By providing an objective, biological measure of injury, molecular markers could help ensure that individuals who sustain a concussion are appropriately managed and protected from further harm.
IN CONCLUSION
The identification of molecular markers for mTBI is poised to transform concussion diagnosis from a largely subjective process into one that is objective and precise. By providing a more accurate and timely assessment of brain injury, molecular markers have the potential to improve patient care, reduce the risk of further injury, and enhance recovery outcomes. While more research is needed to refine these biomarkers and establish standardized testing protocols, the progress made so far brings us closer to the day when definitive concussion diagnosis is a reality. With continued advancements in molecular biology, it may soon be possible to accurately diagnose and manage mTBI with the same level of certainty that we apply to other medical conditions.
Traumatic brain injury (tbi) can have significant and long-lasting effects on communication. Cognitive impairments resulting from TBI can impact an individual’s ability to express thoughts clearly, understand language and engage in social interactions. However, recent advancements in digital communication and technology have opened new pathways to assist individuals with TBI. One of the most promising tools for improving communication in this population is the use of emojis. These small, expressive symbols can help bridge communication gaps, offering a simple, effective means for individuals with TBI to express emotions, convey meaning and engage with others.
UNDERSTANDING TRAUMATIC BRAIN INJURY AND ITS IMPACT ON COMMUNICATION
TBI occurs when a sudden trauma causes damage to the brain, which can result from a fall, accident, sports injury, or violent impact. The effects of TBI vary widely depending on the severity of the injury and the area of the brain affected. Common symptoms include memory loss, difficulty concentrating, emotional instability and problems with speech and language.

For many individuals with TBI, language processing and expression become major challenges. These individuals may struggle to find the right words, organize their thoughts, or engage in fluid conversation. In severe cases, individuals may be unable to communicate at all, relying on nonverbal forms of expression. The resulting isolation and frustration may have a profound impact on the emotional well-being of individuals with TBI, further exacerbating communication difficulties.
THE ROLE OF EMOJIS IN COMMUNICATION
Emojis are small, digital images or icons used to convey emotions, ideas, or reactions in electronic communication. Emojis have become an integral part of texting, social media interactions and online conversations. Their simplicity and visual appeal make them particularly effective for conveying emotions quickly and clearly, making them an ideal tool for individuals with communication challenges.
Emojis help bridge the gap between text-based communication and nonverbal cues which are often lost
in written communication. For individuals with TBI,
using emojis can provide an intuitive, accessible way to communicate their feelings, reactions and thoughts, even when words may fail them. They can add a layer of emotional context to text messages, helping others understand the underlying sentiment behind the words.
HOW EMOJIS BENEFIT INDIVIDUALS WITH TBI
Nonverbal Communication Enhancement: One of the key struggles individuals with TBI face is the difficulty in expressing emotions or conveying the intended tone. In speech and written communication, individuals with TBI may find it challenging to express sadness, joy, anger, or excitement in a way that is easily understood by others. Emojis serve as a visual representation of these emotions, providing a clear and direct way for individuals to express themselves. For example, a simple smiley face can indicate happiness or contentment, while a sad face can convey sadness or frustration. This nonverbal support allows individuals to communicate more effectively, even when their verbal skills are impaired.
Improved Clarity in Communication: TBI can often cause cognitive difficulties, including problems with word-finding, memory and sentence construction. In these instances, emojis may help fill in the gaps, allowing individuals to convey meaning without needing to rely solely on words. For example, if someone struggles to describe an event, they may choose to pair a thumbs-up emoji with a few words, indicating approval or agreement. This visual support enhances the clarity of the message, helping to reduce misunderstandings and ensure effective communication.
participate in conversations. The use of emojis can encourage social interaction by lowering the barriers to communication. Emojis provide a less intimidating way for individuals with TBI to engage in digital conversations. A single emoji can be an easy and approachable way to respond to others, express gratitude, or show interest in a topic, even when verbal communication feels overwhelming or difficult.
Reducing Emotional Frustration: Difficulty communicating effectively can lead to frustration, anxiety and depression for individuals with TBI. The inability to express one’s thoughts and emotions in a clear manner may create feelings of helplessness. Emojis offer a way to quickly express how one feels without the pressure of finding the right words. By using emojis, individuals can feel more in control of their communication, which can reduce feelings of frustration and help them maintain emotional stability.
Supporting Cognitive Rehabilitation: Cognitive rehabilitation is an important part of the recovery process for individuals with TBI. Therapy often focuses on improving cognitive functions such as memory, attention and problem-solving skills. Emojis can play a role in this process by providing
a visual tool to reinforce cognitive exercises. For example, therapists may encourage patients to use specific emojis as part of their rehabilitation program, helping them remember and recall emotions, identify appropriate responses and practice communication in a safe, controlled environment.
Enhancing Digital Literacy: In today’s world, digital communication is an essential part of daily life. For individuals recovering from TBI, gaining comfort and confidence with digital platforms is often an important goal. Learning how to use emojis as part of text-based communication can enhance digital literacy, making it easier for individuals to engage with others on social media, send messages to loved ones and participate in online communities. Emojis serve as a nonverbal tool that complements traditional digital communication, helping individuals feel more confident and capable in their online interactions.
CASE STUDIES AND REAL-WORLD APPLICATIONS
A growing body of research and anecdotal evidence suggests that emojis have a positive impact on communication for individuals with TBI. For example, a study conducted with individuals recovering from TBI found that the use of emojis improved the clarity and emotional tone of their written messages. Participants reported feeling more confident in their ability to express emotions and connect with others. Additionally, caregivers and family members noted that emojis helped reduce the frustration associated with communication difficulties, leading to improved relationships and better emotional well-being.
In another case, therapists working with individuals who had sustained TBIs in accidents or sports injuries integrated emojis into their rehabilitation programs. By using emojis to represent different emotions or actions, individuals were able to engage more effectively with cognitive exercises. The therapists noted that this simple tool helped reinforce lessons about emotional expression, improved social skills and encouraged more interactive communication.
Moreover, families of individuals with TBI have reported that the use of emojis has helped maintain relationships by providing an easier way for loved ones to communicate. Emojis have become an essential tool for those supporting persons with TBI, offering a nonverbal way to check in, offer support, or show empathy without overwhelming the individuals with excessive verbal demands.
IN CONCLUSION
Emojis represent a simple yet powerful tool for individuals with traumatic brain injury to enhance communication, express emotions and reduce social isolation. By offering a visual and intuitive means of communication, emojis help individuals with TBI overcome language barriers, alleviate frustration and engage more effectively with others. As digital communication continues to play an increasingly important role in modern society, the use of emojis can be an essential part of the recovery and rehabilitation process for individuals with TBI. While they may not be a cure-all, emojis provide a meaningful way to support communication and improve quality of life for those affected by TBI, offering a small yet significant step toward a more inclusive and understanding world.
In recent years, few names have been as synonymous with technological innovation as Elon Musk. Known for his groundbreaking ventures such as Tesla and SpaceX, Musk has now turned his attention to the human brain, founding Neuralink — a company that aims to revolutionize the field of neuroscience. Through Neuralink, Musk envisions a world where brain-computer interfaces (BCIs) are no longer a far-off science fiction concept but an accessible reality capable of transforming lives. While the company’s overarching goal is to merge the human brain with artificial intelligence, one of the most promising potential applications lies in its ability to help individuals suffering from traumatic brain injuries (TBI). By using advanced neural interfaces, Neuralink may one day provide life-changing treatments for those whose lives have been upended by severe brain damage.
THE BIRTH OF NEURALINK
Founded in 2016, Neuralink was conceived by Elon Musk with the intention of advancing the development of brain-machine interfaces to solve some of the most pressing problems in human health and consciousness. Musk, a visionary known for pushing the boundaries of technology, recognized that the future of artificial intelligence could pose significant challenges to humanity. His response was to find a way for the human brain to communicate more seamlessly with AI — ensuring that humans could co-exist with intelligent machines rather than be left behind.
Neuralink’s original goal was not just to develop a tool for brain augmentation, but also to treat and manage neurological diseases and injuries. Musk has frequently stated that he believes the integration of the human brain with computers could improve human cognition and potentially lead to the development of new capabilities, such as memory enhancement or increased intelligence. In addition, by creating a direct neural interface, the company could help manage conditions like Alzheimer’s disease, Parkinson’s disease and traumatic brain injury (TBI), ultimately improving the quality of life for millions of people around the world.

At the heart of Neuralink’s technology is a tiny, flexible electrode that can be implanted into the brain. The electrode, which connects to a small device resembling a computer chip, would read and transmit electrical signals from the brain, providing a direct communication channel between the brain and external devices. This could enable a wide range of applications, from controlling prosthetic limbs to restoring lost sensory functions. The implications for treating TBI — where brain tissue is damaged by a blow or jolt to the head — are profound, as Neuralink could offer new solutions to patients who might otherwise have limited options for recovery.
TRAUMATIC BRAIN INJURY: A GROWING GLOBAL EPIDEMIC
TBI is a serious and growing problem worldwide. According to the Centers for Disease Control and Prevention (CDC), TBIs are responsible for over 2.8 million emergency department visits, hospitalizations and deaths in the United States alone each year. These injuries can result from a wide variety of events, including car accidents, sports injuries, falls and physical violence. While some individuals recover from mild TBIs (commonly known as concussions), others experience more severe and long-lasting effects that can impact their cognitive abilities, emotions and motor functions.
In its most severe form, TBI can lead to permanent disability or even death. Even those who survive a traumatic brain injury may suffer lifelong consequences, including memory loss, personality changes, impaired speech, paralysis and other neurological impairments. The economic and emotional toll on patients and their families is staggering, with rehabilitation and ongoing medical care often costing millions of dollars. Despite years of research and medical advancement, effective treatments for traumatic brain injury have remained limited and many patients are left with few options for recovery.
It is in this context that Neuralink’s technology offers a beacon of hope. By developing a sophisticated brain-machine interface, Neuralink has the potential to bridge the gap between the injured brain and modern technology, providing new opportunities for patients with TBIs to regain lost functions and improve their quality of life.
THE SCIENCE BEHIND NEURALINK AND ITS POTENTIAL FOR TBI RECOVERY
At the core of Neuralink’s technology lies a deep understanding of how the brain works. The brain is made up of billions of neurons that communicate with one another through electrical signals. These signals form the basis of all cognitive functions — such as thought, memory, emotion and movement. When the brain is injured, however, these electrical pathways can become disrupted, leading to loss of function and neurological impairments.
Neuralink aims to address this issue by developing a system that can both read and stimulate the brain’s electrical activity. By implanting electrodes into specific regions of the brain, the device would be able to detect abnormal activity associated with TBI and potentially even restore function to damaged areas. The electrodes could also be used to transmit signals to the brain, encouraging the regeneration of neural pathways or bypassing damaged regions altogether.
For patients with TBI, Neuralink’s BCI could be used in several ways to aid recovery. One of the most promising applications is neuroplasticity — the brain’s ability to reorganize itself
by forming new neural connections in response to injury. Neuralink’s electrodes could help stimulate specific regions of the brain, promoting the development of new pathways that could compensate for areas that have been damaged. In essence, the device would act as a form of brain therapy, helping the brain rewire itself and regain lost functions.
In addition to promoting neuroplasticity, Neuralink’s technology could also provide real-time monitoring of brain activity, allowing healthcare providers to gain deeper insights into a patient’s condition. By tracking brainwaves and neural signals, doctors could identify patterns of injury, monitor recovery progress and adjust treatment plans accordingly. For patients with severe TBIs, this could lead to more personalized and effective treatment strategies.
Moreover, Neuralink’s ability to interface with external devices could enable patients to control prosthetic limbs or communicate more effectively, even if their ability to speak or move has been compromised. The technology could provide a direct line of communication between the brain and robotic systems, allowing individuals with TBI to interact with their environment in ways that would otherwise be impossible.

OVERCOMING CHALLENGES AND ETHICAL CONSIDERATIONS
While the potential of Neuralink’s technology is immense, there are still significant challenges to overcome before it can be used to treat traumatic brain injury. One of the biggest hurdles is ensuring the safety and efficacy of the devices. Implanting electrodes into the brain is a delicate procedure that carries inherent risks, including infection, bleeding and damage to surrounding tissue. Additionally, the long-term effects of having such a device implanted in the brain are not yet fully understood.
To address these concerns, Neuralink has conducted animal studies and is working on perfecting its surgical techniques to make the implantation process as minimally invasive as possible. The company has developed a robotic surgical system that can perform the procedure with high precision, reducing the risk of complications. While human trials are still in the early stages, Neuralink has shown promising results in animal models, demonstrating that the technology is capable of recording brain activity and stimulating neural pathways.
There are also ethical considerations surrounding the use of brain-machine interfaces, particularly when it comes to issues of privacy, consent and autonomy. As Neuralink’s technology advances, questions will inevitably arise about who has access to the data generated by the devices and how that information is used. Furthermore, the prospect of enhancing human cognition through neural interfaces raises concerns about inequality and the potential for misuse.
Despite these challenges, the potential benefits of Neuralink’s technology for people with traumatic brain injury cannot be overstated. For individuals whose lives have been profoundly impacted by TBI, the promise of new treatments and improved outcomes offers a glimmer of hope.
A FUTURE OF POSSIBILITIES
The future of Neuralink holds exciting possibilities, not just for those with traumatic brain injuries but for humanity as a whole. By developing advanced BCIs that can repair and enhance brain function, Neuralink could revolutionize the way we understand the human mind and open up new avenues for treating a wide range of neurological disorders.
As research and development continue, it is likely that Neuralink will collaborate with leading neuroscientists, medical professionals and healthcare providers to refine its technology and explore its full potential. In the coming years, we may see the first successful treatments for TBI using Neuralink’s innovative devices, ushering in a new era of brain rehabilitation.

While much work remains to be done, Neuralink represents a bold step toward bridging the gap between science fiction and reality. For those living with the devastating effects of traumatic brain injury, the dream of recovery may no longer be a distant hope but a real and achievable future.
In the hands of Elon Musk and his team at Neuralink, the human brain may one day be equipped with the tools it needs to heal, adapt and thrive — ushering in a new era of neurological treatment and opening the door to a world where the impossible becomes possible.
Despite these challenges, the potential benefits of Neuralink’s technology for people with traumatic brain injury cannot be overstated. For individuals whose lives have been profoundly impacted by TBI, the promise of new treatments and improved outcomes offers a glimmer of hope.
A FUTURE OF POSSIBILITIES
The future of Neuralink holds exciting possibilities, not just for those with traumatic brain injuries but for humanity as a whole. By developing advanced BCIs that can repair and enhance brain function, Neuralink could revolutionize the way we understand the human mind and open up new avenues for treating a wide range of neurological disorders.
As research and development continue, it is likely that Neuralink will collaborate with leading neuroscientists, medical professionals and healthcare providers to refine its technology and explore its full potential. In the coming years, we may see the first successful treatments for TBI using Neuralink’s innovative devices, ushering in a new era of brain rehabilitation.
While much work remains to be done, Neuralink represents a bold step toward bridging the gap between science fiction and reality. For those living with the devastating effects of traumatic brain injury, the dream of recovery may no longer be a distant hope but a real and achievable future.
In the hands of Elon Musk and his team at Neuralink, the human brain may one day be equipped with the tools it needs to heal, adapt and thrive — ushering in a new era of neurological treatment and opening the door to a world where the impossible becomes possible.
Polytrauma is a specific medical term that describes the condition of someone who has sustained injuries to multiple body parts and organ systems. For instance, in a car crash, an individual may suffer serious burns over large portions of his or her body in addition to broken bones or a traumatic brain injury (TBI). The critical nature of an injury is evaluated in the U.S. medical community through a scale referred to as the Injury Severity Score (ISS). Polytraumas have scores of 16 or greater on this scale.
CAUSES OF POLYTRAUMA
Motor vehicle accidents are a major cause of polytrauma among civilians. The high speeds and sudden impacts that occur in many types of car crashes often lead to disastrous outcomes. The U.S. military has used the medical designation of polytrauma to categorize injuries sustained by military personnel during conflicts. Military personnel are subject to some of the most serious threats of polytrauma injuries, including blasts from explosive devices.
Beyond serious motor vehicle accidents and military-related incidents, causes of polytrauma can also include knife wounds, physical altercations, gunshot wounds, fire-related injuries, and falls from elevated heights. Polytrauma often involves a combination of TBI with other debilitating injuries such as serious eye damage, hearing damage, amputations, spinal cord injury, and severe burns. In addition, some individuals experience post-traumatic stress disorder (PTSD). Historically, polytrauma survival rates have been low, however, with advances in medical technology these statistics have greatly improved.

TREATMENT OF POLYTRAUMA PATIENTS WITH TBI
In medical terms, the presence of TBI in a polytrauma patient is referred to as Polytrauma and Concomitant Traumatic Brain Injury. According to the Journal of Neurosurgery, clinicians face unique challenges from decision-making and rehabilitative perspective when it comes to the treatment of these patients. Treatment management is complex and should be systematic, beginning at the scene with timely transport. Early operative interventions are also key.
According to the World Journal of Emergency Surgery, critical trauma care is ever-improving, yet TBI-related mortality rates are rising compared to other causes of death. Managing the acute phase after a severe TBI with polytrauma represents a challenging situation for every trauma team member and often involves the ‘damage control’ approach to sustain life. The challenge with polytrauma and concomitant traumatic brain injury patients is making sure equal emphasis is placed on stabilizing life-sustaining systems and doing everything possible to minimize brain damage.
POLYTRAUMA COULD WORSEN BRAIN INJURY
Primary brain injury results from mechanical injury at the time of the trauma whereas secondary brain injury is caused by the physiologic responses to the initial injury. Post-trauma care, as it relates to traumatic brain injury, is focused on halting or minimizing bleeding or clotting in the brain, among other biochemical processes, all of which result in secondary brain injury. Because polytrauma patients may require multiple damage control procedures, there is a risk of not providing ample emphasis on brain care. In addition, the presence of hypotension (low blood pressure), hypoxia (low oxygen levels), and fever — all commonly found in polytrauma patients — have been shown to initiate secondary brain damage.
TRAUMA NEUROSURGERY
The trauma neurosurgeon plays a key role in being able to ensure optimal treatment of polytrauma and concomitant TBI patients while minimizing secondary brain damage. Although brain surgery is highly complex and specialized, how it helps TBI patients comes down to decompression. Known as a craniotomy, this surgery relieves pressure on the brain, in turn slowing secondary brain damage. Dating back to Hippocrates, who is documented to have suggested their use in treating TBI, craniotomy revolutionized neurosurgery.
CHOOSING THE BEST TRAUMA CENTER
The best facilities for polytrauma patients to receive care are level one trauma centers. According to the American Trauma Society, A level one trauma center is capable of providing total care for every aspect of injury from prevention through rehabilitation, including surgical intervention. Although a level one trauma center can provide comprehensive treatment to trauma victims, they are not always capable of providing what is known as Simultaneous Multisystem Surgery (SMS) — which allows for different surgical teams to work on a patient simultaneously — the key to optimally treating polytrauma patients with TBI.
For a level one trauma center to provide SMS they must be equipped with a hybrid-emergency room system (HERS) where diagnostic procedures, such as CT scans, and damage control interventions, such as neurosurgery, can be performed simultaneously without patient transfer. Studies have shown that the HERS approach has been associated with a shorter time to initiate CT scanning, emergency surgery, and fewer unfavorable outcomes in polytrauma patients with and without TBI — ultimately resulting in higher – and more functional — survival rates.
While the ability to perform SMS seems to be straightforward and beneficial, very few centers in the United States are equipped with HERS and have trained staff to perform SMS in the context of TBI complicated by polytrauma. Other countries appear to be ahead of the curve in this area of trauma response. For example, not only are many of Japan’s trauma centers equipped with HERS, but they have also developed the Japanese Association for Hybrid Emergency Room Systems to specifically address the needs of specialized HERS trauma teams.
FACTORS TO CONSIDER FOR THE BEST OUTCOME
According to an article published in the National Library
of Medicine, the occurrence of TBI in polytrauma patients increases mortality and reduces their quality of life. Studies have shown that the crucial factors for ensuring the best outcomes in these cases involve getting the most appropriate care as quickly as possible. Factors such as the speed in which ambulatory care is on scene and the patient being transported to a level one trauma center — when possible one equipped to administer SMS — can make substantial differences in a patient’s recovery.
The World Health Organization estimates that traumatic brain injury (TBI) is and will remain the most important cause of neurodisability in the coming years. The search for neuroprotective therapies for severe TBI has been extensive but unfruitful over the last few decades, testified by mo e than 30 failed clinical trials, and we still have no specific neu oprotective therapy, that is, effective in clinica TBI. The burden of mortality and residual disability calls for new approaches to promote recovery of function of TBI patients in the acute and chronic phase.
Classically described as a sudden event with short-term consequences, TBI induces dynamic pathological cascades that may persist for months or years after injury with a major impact on outcome. Among dynamic mechanisms, the neuroinflammatory esponse and the accumulation of aberrant proteins may have a critical role in establishing a neuropathological link between acute mechanical injury and late neurodegeneration. The close association between post-TBI neurological changes, persistent neuroinflammation, and lat neuropathology highlights the fact that the window of opportunity for therapeutic intervention may be much wider than previously thought and that long-term treatment encompassing the acute and chronic phase should be tested to effectively interfe e with this complex condition.
An emerging technology, virtual reality (VR), represents a new tool for this purpose and might provide TBI care teams with new neuro-restorative strategies readily available at the bedside. Since the late 1980s, this term has been used to describe a 3D synthetic environment created by computer graphics, where the user has the feeling of being inside. VR can be described as “an advanced form of human-computer interface that allows the user to interact with and become immersed in a computer-generated environment in a naturalistic fashion”. For its flexibilit , sense of presence (i.e., the feeling of “being there”) and emotional engagement, VR has been tested in motor and cognitive rehabilitation, with good results. In stroke patients, the number of VR programs is rapidly increasing with compelling data showing an improvement in recovery of motor function and daily living activities. VR has been successfully used both as assessment instrument and as therapeutic intervention. As assessment tool, VR has been used to detect visual-vestibular deficits in adults after concussion and mild BI. VR assessment protocols appear to be primarily implemented for mild TBI. Conversely, VR treatment protocols for cognitive rehabilitation are used transversely from mild to severe conditions, although effectiveness of thes kinds of interventions needs to be further explored.
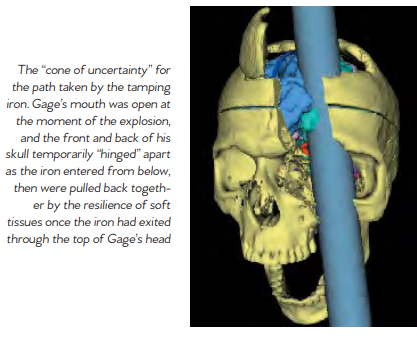
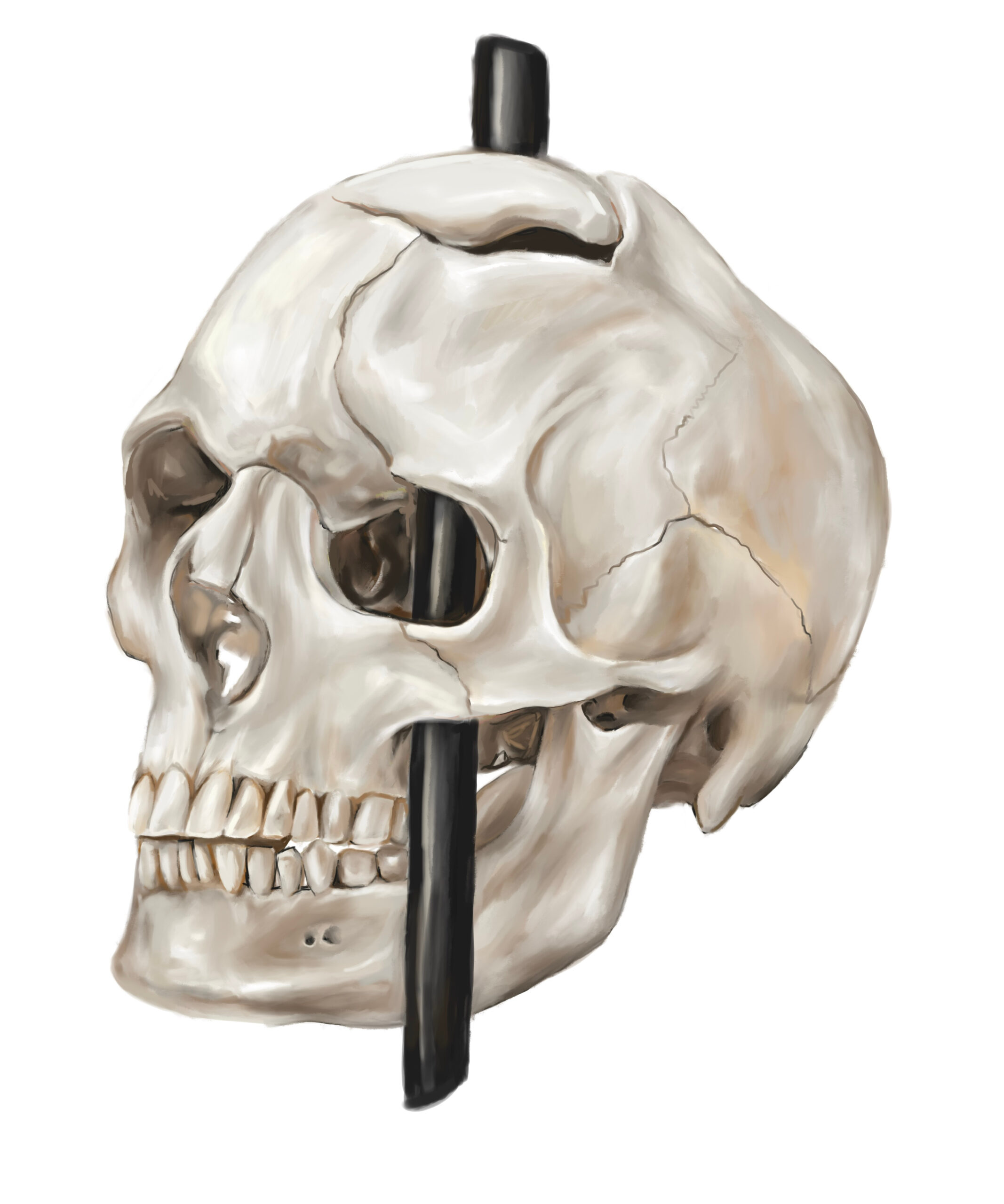
Phineas P. Gage (1823–1860) was an American railroad construction foreman remembered for his improbable survival of an accident in which a large iron rod was driven completely through his head, destroying much of his brain’s left frontal lobe, and for that injury’s reported effects on his personality and behavior over the remaining 12 years of his life. Long known as the “American Crowbar Case”—once termed “the case which more than all others is calculated to excite our wonder, impair the value of prognosis, and even to subvert our physiological doctrines” —Phineas Gage influenced 19t -century discussion about the mind and brain, particularly debate on cerebral localization, and was perhaps the first case to suggest the brain’s ole in determining personality, and that damage to specific parts of the brain might induce specific mental changes I n 1848, Gage, 25, was the foreman of a crew cutting a railroad bed in Cavendish, Vermont. On September 13, as he was using a tamping iron to pack explosive powder into a hole, the powder detonated. The tamping iron—43 inches long, 1.25 inches in diameter and weighing 13.25 pounds— shot skyward, penetrated Gage’s left cheek, ripped into his brain and exited through his skull, landing several dozen feet away. Though blinded in his left eye, he might not even have lost consciousness, and he remained savvy enough to tell a doctor that day, “Here is business enough for you.”
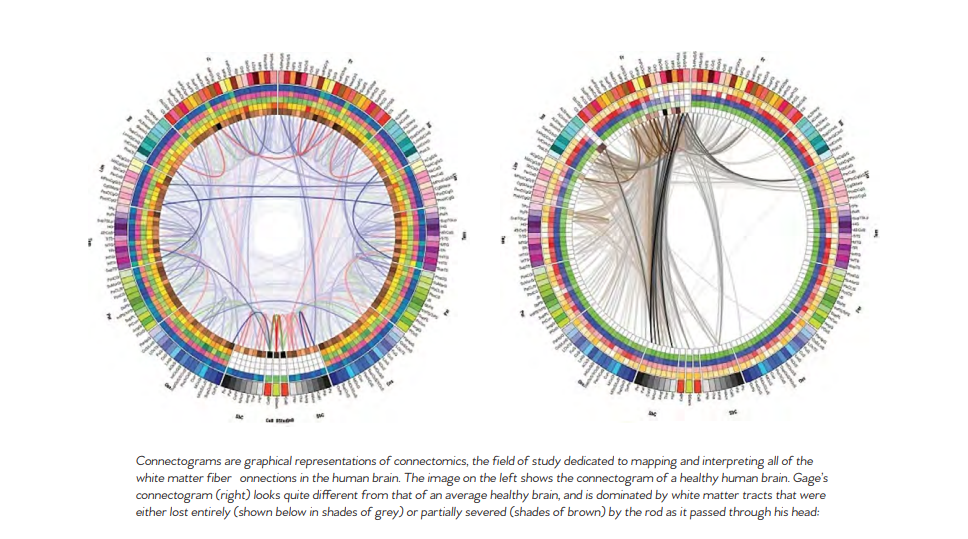
Gage’s initial survival would have ensured him a measure of celebrity, but his name was etched into history by observations made by John Martyn Harlow, the doctor who treated him for a few months afterward. Gage’s friends found him“no longer Gage,” Harlow wrote. The balance between his “intellectual faculties and animal propensities” seemed gone. He could not stick to plans, uttered “the grossest profanity” and showed “little deference for his fellows.” The railroad-construction company that employed him, which had thought him a model foreman, refused to take him back. So Gage went to work at a stable in New Hampshire, drove coaches in Chile and eventually joined relatives in San Francisco, where he died in May 1860, at age 36, after a series of seizures. In time, Gage became the most famous patient in the annals of neuroscience, because his case was the first to suggest a link between brain trauma and personality change. In his book An Odd Kind of Fame: Stories of Phineas Gage, the University of Melbourne’s Malcolm Macmillan writes that two-thirds of introductory psychology textbooks mention Gage. Even today, his skull, the tamping iron and a mask of his face made while he was alive are the most sought-out items at the Warren Anatomical Museum on the Harvard Medical School campus. Michael Spurlock, a database administrator in Missoula, Montana, happened upon the Wilgus daguerreotype on Flickr in December 2008. As soon as he saw the object the one-eyed man held, Spurlock knew it was not a harpoon. Too short. No wooden shaft. It looked more like a tamping iron, he thought. Instantly, a name popped into his head: Phineas Gage. Spurlock knew the Gage story well enough to know that any photograph of him would be the first to come to light. He knew enough, too, to be intrigued by Gage’s appearance, if it was Gage. Over the years, accounts of his changed character had gone far beyond Harlow’s observations, Macmillan says, turning him into an ill-tempered, shiftless drunk. But the man in the Flickr photogragh seemed well-dressed and confident It was Spurlock who told the Wilguses that the man in their daguerreotype might be Gage. After Beverly finished her online research, she and Jack concluded that the man probably was. She e-mailed a scan of the photograph to the Warren museum. Eventually it reached Jack Eckert, the public-services librarian at Harvard’s Center for the History of Medicine. “Such a ‘wow’ moment,” Eckert recalls. It had to be Gage, he determined. How many mid-19th-century men with a mangled eye and scarred forehead had their portrait taken holding a metal tool? A tool with an inscription on it? The Wilguses had never noticed the inscription; after all, the daguerreotype measures only 2.75 inches by 3.25 inches. But a few days after receiving Spurlock’s tip, Jack, a retired photography professor, was focusing a camera to take a picture of his photograph. “There’s writing on that rod!” Jack said. He couldn’t read it all, but part of it seemed to say, “through the head of Mr. Phi...”
In March 2009, Jack and Beverly went to Harvard to compare their picture with Gage’s mask and the tamping iron, which had been inscribed in Gage’s lifetime: “This is the bar that was shot through the head of Mr. Phinehas P. Gage,” it reads, misspelling the name. Harvard has not officially decl ed that the daguerreotype is of Gage, but Macmillan, whom the Wilguses contacted next, is quite certain. He has also learned of another photograph, he says, kept by a descendant of Gage’s. As for Spurlock, when he got word that his hunch was apparently correct, “I threw open the hallway door and told my wife, ‘I played a part in a historical discovery!’ ” Popular reports of Gage often depict him as a hardworking, pleasant man prior to the accident. Post-accident, these reports describe him as a changed man, suggesting that the injury had transformed him into a surly, aggressive heavy drinker who was unable to hold down a job. Harlow presented the first account of the changes in Gage’s behavior following the accident. Where Gage had been described as energetic, motivated, and shrewd prior to the accident, many of his acquaintances explained that after the injury he was “no longer Gage.” Since there is little direct evidence of the exact extent of Gage’s injuries aside from Harlow’s report, it is difficult to know exact how severely his brain was damaged. Harlow’s accounts suggest that the injury did lead to a loss of social inhibition, leading Gage to behave in ways that were seen as inappropriate. Gage’s case had a tremendous influence on early neu ology. The specific changes observed in his behavior pointed t emerging theories about the localization of brain function, or the idea that certain functions are associated with specific a eas of the brain. In those years, neurology was in its infancy. Gage’s extraordinary story served as one of the first sou ces of evidence
that the frontal lobe was involved in personality. Today, scientists better understand the role that the frontal cortex has to play in important higher-order functions such as reasoning, language, and social cognition. After the accident, Gage was unable to continue his previous job. According to Harlow, Gage spent some time traveling through New England and Europe with his tamping iron to earn money, supposedly even appearing in the Barnum American Museum in New York. He also worked briefly at a livery stabl in New Hampshire and then spent seven years as a stagecoach driver in Chile. He eventually moved to San Francisco to live with his mother as his health deteriorated. After a series of epileptic seizures, Gage died on May 21, 1860, almost 12 years after his accident. Seven years later, Gage’s body was exhumed. His brother gave his skull and the tamping rod to Dr. Harlow, who subsequently donated them to the Harvard University School of Medicine. They are still exhibited in its museum today.
Contribute to the TBI Times