In recent years, few names have been as synonymous with technological innovation as Elon Musk. Known for his groundbreaking ventures such as Tesla and SpaceX, Musk has now turned his attention to the human brain, founding Neuralink — a company that aims to revolutionize the field of neuroscience. Through Neuralink, Musk envisions a world where brain-computer interfaces (BCIs) are no longer a far-off science fiction concept but an accessible reality capable of transforming lives. While the company’s overarching goal is to merge the human brain with artificial intelligence, one of the most promising potential applications lies in its ability to help individuals suffering from traumatic brain injuries (TBI). By using advanced neural interfaces, Neuralink may one day provide life-changing treatments for those whose lives have been upended by severe brain damage.
THE BIRTH OF NEURALINK
Founded in 2016, Neuralink was conceived by Elon Musk with the intention of advancing the development of brain-machine interfaces to solve some of the most pressing problems in human health and consciousness. Musk, a visionary known for pushing the boundaries of technology, recognized that the future of artificial intelligence could pose significant challenges to humanity. His response was to find a way for the human brain to communicate more seamlessly with AI — ensuring that humans could co-exist with intelligent machines rather than be left behind.
Neuralink’s original goal was not just to develop a tool for brain augmentation, but also to treat and manage neurological diseases and injuries. Musk has frequently stated that he believes the integration of the human brain with computers could improve human cognition and potentially lead to the development of new capabilities, such as memory enhancement or increased intelligence. In addition, by creating a direct neural interface, the company could help manage conditions like Alzheimer’s disease, Parkinson’s disease and traumatic brain injury (TBI), ultimately improving the quality of life for millions of people around the world.

At the heart of Neuralink’s technology is a tiny, flexible electrode that can be implanted into the brain. The electrode, which connects to a small device resembling a computer chip, would read and transmit electrical signals from the brain, providing a direct communication channel between the brain and external devices. This could enable a wide range of applications, from controlling prosthetic limbs to restoring lost sensory functions. The implications for treating TBI — where brain tissue is damaged by a blow or jolt to the head — are profound, as Neuralink could offer new solutions to patients who might otherwise have limited options for recovery.
TRAUMATIC BRAIN INJURY: A GROWING GLOBAL EPIDEMIC
TBI is a serious and growing problem worldwide. According to the Centers for Disease Control and Prevention (CDC), TBIs are responsible for over 2.8 million emergency department visits, hospitalizations and deaths in the United States alone each year. These injuries can result from a wide variety of events, including car accidents, sports injuries, falls and physical violence. While some individuals recover from mild TBIs (commonly known as concussions), others experience more severe and long-lasting effects that can impact their cognitive abilities, emotions and motor functions.
In its most severe form, TBI can lead to permanent disability or even death. Even those who survive a traumatic brain injury may suffer lifelong consequences, including memory loss, personality changes, impaired speech, paralysis and other neurological impairments. The economic and emotional toll on patients and their families is staggering, with rehabilitation and ongoing medical care often costing millions of dollars. Despite years of research and medical advancement, effective treatments for traumatic brain injury have remained limited and many patients are left with few options for recovery.
It is in this context that Neuralink’s technology offers a beacon of hope. By developing a sophisticated brain-machine interface, Neuralink has the potential to bridge the gap between the injured brain and modern technology, providing new opportunities for patients with TBIs to regain lost functions and improve their quality of life.
THE SCIENCE BEHIND NEURALINK AND ITS POTENTIAL FOR TBI RECOVERY
At the core of Neuralink’s technology lies a deep understanding of how the brain works. The brain is made up of billions of neurons that communicate with one another through electrical signals. These signals form the basis of all cognitive functions — such as thought, memory, emotion and movement. When the brain is injured, however, these electrical pathways can become disrupted, leading to loss of function and neurological impairments.
Neuralink aims to address this issue by developing a system that can both read and stimulate the brain’s electrical activity. By implanting electrodes into specific regions of the brain, the device would be able to detect abnormal activity associated with TBI and potentially even restore function to damaged areas. The electrodes could also be used to transmit signals to the brain, encouraging the regeneration of neural pathways or bypassing damaged regions altogether.
For patients with TBI, Neuralink’s BCI could be used in several ways to aid recovery. One of the most promising applications is neuroplasticity — the brain’s ability to reorganize itself
by forming new neural connections in response to injury. Neuralink’s electrodes could help stimulate specific regions of the brain, promoting the development of new pathways that could compensate for areas that have been damaged. In essence, the device would act as a form of brain therapy, helping the brain rewire itself and regain lost functions.
In addition to promoting neuroplasticity, Neuralink’s technology could also provide real-time monitoring of brain activity, allowing healthcare providers to gain deeper insights into a patient’s condition. By tracking brainwaves and neural signals, doctors could identify patterns of injury, monitor recovery progress and adjust treatment plans accordingly. For patients with severe TBIs, this could lead to more personalized and effective treatment strategies.
Moreover, Neuralink’s ability to interface with external devices could enable patients to control prosthetic limbs or communicate more effectively, even if their ability to speak or move has been compromised. The technology could provide a direct line of communication between the brain and robotic systems, allowing individuals with TBI to interact with their environment in ways that would otherwise be impossible.

OVERCOMING CHALLENGES AND ETHICAL CONSIDERATIONS
While the potential of Neuralink’s technology is immense, there are still significant challenges to overcome before it can be used to treat traumatic brain injury. One of the biggest hurdles is ensuring the safety and efficacy of the devices. Implanting electrodes into the brain is a delicate procedure that carries inherent risks, including infection, bleeding and damage to surrounding tissue. Additionally, the long-term effects of having such a device implanted in the brain are not yet fully understood.
To address these concerns, Neuralink has conducted animal studies and is working on perfecting its surgical techniques to make the implantation process as minimally invasive as possible. The company has developed a robotic surgical system that can perform the procedure with high precision, reducing the risk of complications. While human trials are still in the early stages, Neuralink has shown promising results in animal models, demonstrating that the technology is capable of recording brain activity and stimulating neural pathways.
There are also ethical considerations surrounding the use of brain-machine interfaces, particularly when it comes to issues of privacy, consent and autonomy. As Neuralink’s technology advances, questions will inevitably arise about who has access to the data generated by the devices and how that information is used. Furthermore, the prospect of enhancing human cognition through neural interfaces raises concerns about inequality and the potential for misuse.
Despite these challenges, the potential benefits of Neuralink’s technology for people with traumatic brain injury cannot be overstated. For individuals whose lives have been profoundly impacted by TBI, the promise of new treatments and improved outcomes offers a glimmer of hope.
A FUTURE OF POSSIBILITIES
The future of Neuralink holds exciting possibilities, not just for those with traumatic brain injuries but for humanity as a whole. By developing advanced BCIs that can repair and enhance brain function, Neuralink could revolutionize the way we understand the human mind and open up new avenues for treating a wide range of neurological disorders.
As research and development continue, it is likely that Neuralink will collaborate with leading neuroscientists, medical professionals and healthcare providers to refine its technology and explore its full potential. In the coming years, we may see the first successful treatments for TBI using Neuralink’s innovative devices, ushering in a new era of brain rehabilitation.

While much work remains to be done, Neuralink represents a bold step toward bridging the gap between science fiction and reality. For those living with the devastating effects of traumatic brain injury, the dream of recovery may no longer be a distant hope but a real and achievable future.
In the hands of Elon Musk and his team at Neuralink, the human brain may one day be equipped with the tools it needs to heal, adapt and thrive — ushering in a new era of neurological treatment and opening the door to a world where the impossible becomes possible.
Despite these challenges, the potential benefits of Neuralink’s technology for people with traumatic brain injury cannot be overstated. For individuals whose lives have been profoundly impacted by TBI, the promise of new treatments and improved outcomes offers a glimmer of hope.
A FUTURE OF POSSIBILITIES
The future of Neuralink holds exciting possibilities, not just for those with traumatic brain injuries but for humanity as a whole. By developing advanced BCIs that can repair and enhance brain function, Neuralink could revolutionize the way we understand the human mind and open up new avenues for treating a wide range of neurological disorders.
As research and development continue, it is likely that Neuralink will collaborate with leading neuroscientists, medical professionals and healthcare providers to refine its technology and explore its full potential. In the coming years, we may see the first successful treatments for TBI using Neuralink’s innovative devices, ushering in a new era of brain rehabilitation.
While much work remains to be done, Neuralink represents a bold step toward bridging the gap between science fiction and reality. For those living with the devastating effects of traumatic brain injury, the dream of recovery may no longer be a distant hope but a real and achievable future.
In the hands of Elon Musk and his team at Neuralink, the human brain may one day be equipped with the tools it needs to heal, adapt and thrive — ushering in a new era of neurological treatment and opening the door to a world where the impossible becomes possible.
Axolotls, a species of salamanders native to lakes near Mexico City, have long captivated the scientific community due to their extraordinary regenerative abilities. These amphibians can regenerate entire limbs, spinal cord, heart and even parts of their brain. However, the molecular mechanisms that allow such remarkable tissue regrowth have remained a subject of intense study. A groundbreaking new technique, the first-ever axolotl stereo-seq, is shedding new light on these regenerative processes, especially in the brain. This pioneering method holds tremendous promise for advancing our understanding of neural regeneration and potentially developing new therapies for neurodegenerative diseases in humans.
WHAT IS STEREO-SEQ?
Stereo-seq (short for stereoscopic sequencing) is a cutting-edge technique that allows researchers to map gene expression with high spatial resolution. In essence, it is a method for visualizing the activity of genes within the context of tissue architecture. By capturing both the spatial and transcriptomic data of cells, stereo-seq offers unparalleled insights into how gene expression varies across different regions of a tissue or organ.
Traditional gene expression studies, such as RNA sequencing, can only give a broad view of gene activity at the molecular level, without considering the exact location of that activity within the tissue. Stereo-seq bridges this gap by combining high-resolution spatial tissue imaging with gene sequencing, creating a comprehensive map that reveals not only what genes are active but also where they are active.

The axolotl is a paedomorphic salamander closely related to the tiger salamander. It is unusual among amphibians in that it reaches adulthood without undergoing metamorphosis.
THE AXOLOTL: A MODEL ORGANISM FOR REGENERATION
The axolotl has become one of the most important model organisms in regenerative biology. Unlike most vertebrates, axolotls possess the incredible ability to regenerate complex body parts, including entire limbs, organs and even parts of their brain. This ability has made axolotls an ideal subject for studying regeneration at a molecular level.
In particular, the axolotl’s brain offers an exciting area of study. While mammals, including humans, have limited capacity to regenerate brain cells after injury, axolotls can repair and regrow significant portions of their central nervous system. Understanding how axolotls are able to achieve this could revolutionize the treatment of neurodegenerative conditions such as Alzheimer’s, Parkinson’s and multiple sclerosis.
THE ROLE OF BRAIN CELL REGENERATION IN AXOLOTLS
One of the most intriguing aspects of axolotl regeneration is their ability to regenerate brain cells after injury. In mammals, brain injuries often result in permanent damage because the neurons cannot regrow or repair themselves. However, axolotls have specialized cells, called radial glial cells, that play a crucial role in neurogenesis (the creation of new neurons) in the adult brain.
These radial glial cells are capable of reprogramming themselves to become neuronal precursor cells, which can differentiate into neurons. This process is fundamental to the axolotl’s ability to regenerate parts of its brain. However, the exact molecular signals and genetic pathways that control this process are still poorly understood.
The axolotl is a paedomorphic salamander closely related to the tiger salamander. It is unusual among amphibians in that it reaches adulthood without undergoing metamorphosis.
UNDERSTANDING BRAIN CELL REGENERATION THROUGH STEREO-SEQ
The introduction of stereo-seq to the study of axolotl brain regeneration has allowed scientists to examine the precise molecular events that occur during this process. By applying stereo-seq to axolotl brain tissue, researchers can map gene expression patterns in individual cells while simultaneously preserving the spatial context of the tissue. This enables them to see which genes are active in specific regions of the brain and, more importantly, which genes are involved in the regenerative processes that allow axolotls to regrow brain cells.
The breakthrough stereo-seq study on axolotls focused on several key aspects of brain regeneration, such as:
Cellular Identity and Differentiation: By capturing the gene expression of individual brain cells, researchers were able to identify distinct populations of cells involved in regeneration. This includes understanding the role of radial glial cells and how they differentiate into neurons during brain repair.
Molecular Pathways: Stereo-seq allowed scientists to pinpoint which molecular pathways are activated in response to brain injury. For example, certain genes related to inflammation, cell proliferation and cell differentiation were found to be upregulated in response to injury, suggesting they play a role in the brain’s regenerative process.
Spatial Mapping of Gene Expression: By mapping gene activity in precise spatial locations, researchers could see how different regions of the brain respond to injury. This is critical for understanding how axolotls can regenerate specific brain areas and how this knowledge can be applied to human medicine.
The Stereo-seq Approach, a New Era of Cellular Mapping:
The stereo-seq technique offers several advantages over previous methods used to study tissue regeneration.
HIGH SPATIAL RESOLUTION
One of the biggest challenges in studying tissue regeneration is capturing the precise location of gene activity within a complex tissue structure. Traditional methods, such as bulk RNA sequencing, provide valuable data on gene expression but lack spatial resolution. With stereo-seq, researchers can pinpoint the exact locations within the tissue where specific genes are active. This is particularly important when studying tissues like the brain, where cellular organization is critical to understanding function and regeneration.
INTEGRATION OF SPATIAL AND TRANSCRIPTOMIC DATA
Stereo-seq integrates both spatial and transcriptomic data, allowing researchers to simultaneously visualize the architecture of the tissue and understand the molecular mechanisms at play. This combination provides a much richer and more complete picture of the regeneration process. By studying axolotl brain tissue with this integrated approach, scientists can map out the spatial distribution of cellular activities during regeneration and link them to specific molecular events.
SINGLE-CELL RESOLUTION
Stereo-seq operates at a single-cell resolution, which is essential for understanding the heterogeneity of cells involved in regeneration. In the brain, different types of neurons, glial cells and precursor cells may exhibit different gene expression patterns depending on their developmental stage or regenerative status. Stereo-seq allows researchers to investigate these differences at the level of individual cells, revealing a detailed picture of how various cell types contribute to brain repair.
NEW INSIGHTS INTO AXOLOTL BRAIN REGENERATION
The stereo-seq findings from axolotls have already provided several exciting insights into the regenerative processes at play in their brains:
1. Radial Glial Cells as Key Players in NeurogenesisThe study confirmed that radial glial cells are central to brain cell regeneration in axolotls. These cells, which are typically found in the developing nervous system, retain their regenerative potential in adulthood. Stereo-seq data revealed that radial glial cells not only act as a source of new neurons but also help maintain the architecture of the regenerating tissue.
Interestingly, the study also found that radial glial cells in axolotls exhibit gene expression patterns that are very different from those of radial glial cells in mammals, suggesting that there may be distinct molecular mechanisms at play in the axolotl’s regenerative response.
2. Activation of Regenerative Pathways
Researchers identified several molecular pathways that are upregulated during brain regeneration in axolotls. Some of these pathways are involved in cell proliferation, differentiation and tissue remodeling. These pathways include the Notch, Wnt and Hedgehog signaling pathways, which are known to play crucial roles in development and regeneration in other species as well.
Stereo-seq revealed how these pathways interact in different regions of the brain and how they help guide the regeneration process. By identifying these pathways in axolotls, researchers may be able to design targeted therapies that could encourage neurogenesis in humans, potentially offering new treatments for neurodegenerative diseases.
3. The Importance of Inflammatory ResponseAnother interesting finding from the stereo-seq study was the role of inflammation in brain regeneration. While inflammation is often considered detrimental to tissue repair, the data revealed that a controlled inflammatory response is crucial for promoting neurogenesis in axolotls. This suggests that inflammation may have a dual role, both aiding in the repair of damaged tissue and helping to guide the regenerative process.
IMPLICATIONS FOR HUMAN MEDICINE
The insights gained from studying axolotl brain regeneration using stereo-seq have significant implications for human medicine, particularly in the fields of neurology and regenerative medicine. By understanding the genetic and molecular mechanisms behind axolotl brain repair, scientists can identify potential therapeutic targets for encouraging brain cell regeneration in humans.
IN CONCLUSION
The first-ever axolotl stereo-seq represents a major leap forward in our understanding of brain cell regeneration. By combining spatial mapping with gene expression data, this technique has provided unprecedented insights into the molecular and cellular processes that drive axolotl brain repair. The findings from this study not only deepen our understanding of regenerative biology but also offer exciting new possibilities for treating brain injuries and neurodegenerative diseases in humans. As research in this field continues, we may one day be able to harness the regenerative powers of axolotls to help heal human brains.
Traumatic brain injury (TBI) is a major public health issue that affects millions of people worldwide every year. It results from external forces applied to the head, such as falls, motor vehicle accidents or sports injuries. While the immediate effects of TBI, such as physical injury or cognitive impairment, are well-documented, increasing evidence suggests that TBI can have long-term consequences, including an increased risk for dementia. This article explores the connection between TBI and dementia, the mechanisms that underlie this association, the types of dementia related to TBI and what can be done to mitigate these risks.
UNDERSTANDING TRAUMATIC BRAIN INJURY (TBI)
TBI occurs when a sudden external force causes damage to the brain, either through a direct blow or rapid acceleration or deceleration of the brain inside the skull. TBI can range from mild (concussion) to severe, with varying degrees of recovery. The severity of the injury depends on several factors, including the force of impact, the area of the brain affected and the individual’s health prior to the injury.
Concussions, often referred to as mild TBIs, can cause short-term confusion, dizziness, headache and other symptoms that typically resolve with rest and time. However, even mild TBIs can have long-term consequences if not properly managed, particularly when individuals experience repeated injuries. Severe TBIs can lead to long-lasting impairments in cognitive, physical and emotional functions.

WHAT IS DEMENTIA?
Dementia is a broad term used to describe a decline in cognitive function severe enough to interfere with daily life. It is not a specific disease but a syndrome that may result from various underlying conditions. The most common type of dementia is Alzheimer’s disease, which accounts for up to 70% of all cases. Other forms of dementia include vascular dementia, frontotemporal dementia and Lewy body dementia.
Dementia is characterized by a gradual decline in memory, thinking, judgment and other cognitive abilities. It can also cause behavioral changes, mood disturbances and difficulty performing everyday tasks. The underlying causes of dementia vary but often involve damage to brain cells and the progressive loss of neural connections.
THE LINK BETWEEN TBI AND DEMENTIA
Research has consistently shown that individuals who experience TBI are at a significantly higher risk of developing dementia later in life. This association has been particularly well-documented in people who sustain moderate to severe brain injuries, but even mild TBIs, particularly those involving repeated concussions, have been shown to contribute to an increased risk of dementia.
The connection between TBI and dementia is complex and involves several mechanisms. Key factors include the structural changes in the brain that occur as a result of the injury, the neurochemical processes that are disrupted and the long-term effects of inflammation and cellular damage.
MECHANISMS LINKING TBI AND DEMENTIA
Neuroinflammation: After a TBI, the brain undergoes an inflammatory response as part of the healing process. This inflammation, however, may become chronic, leading to the long-term accumulation of neuroinflammation. Chronic inflammation can accelerate the degeneration of neurons, impair neural communication and promote the formation of neurotoxic proteins such as amyloid-beta and tau. These proteins are hallmark features of Alzheimer’s disease and other forms of dementia.
Amyloid Plaques and Tau Tangles: One of the key pathophysiological features of dementia, particularly Alzheimer’s disease, is the buildup of amyloid plaques and tau tangles in the brain. Amyloid plaques consist of clumps of amyloid-beta protein, while tau tangles involve the abnormal accumulation of tau protein inside neurons. These plaques and tangles disrupt normal brain function and are thought to play a critical role in cognitive decline. Research suggests that TBIs may increase the production of amyloid-beta and tau proteins, making individuals more susceptible to dementia. Studies have found that people with a history of TBI have a higher likelihood of developing these pathological features, which can lead to Alzheimer’s disease and other dementias.
Neuronal Damage and Cell Death: TBI often results in direct damage to brain cells, particularly neurons, which may die or become damaged in ways that impair their function. Over time, this neuronal loss can accumulate and lead to cognitive deficits. In particular, damage to regions of the brain associated with memory, such as the hippocampus, can contribute to the development of dementia.
Disrupted Brain Connectivity: The brain’s networks of neurons are essential for cognitive function. TBIs can disrupt these networks, impairing the brain’s ability to communicate effectively. This disruption may result in long-term cognitive impairments that resemble the early stages of dementia. Over time, these impairments can worsen, leading to the development of neurodegenerative diseases.
Oxidative Stress: Another mechanism that links TBI and dementia is oxidative stress. After an injury, the brain may experience an imbalance between free radicals and antioxidants, leading to oxidative damage to brain cells. This damage can impair cellular function and accelerate neurodegeneration, increasing the risk of dementia.
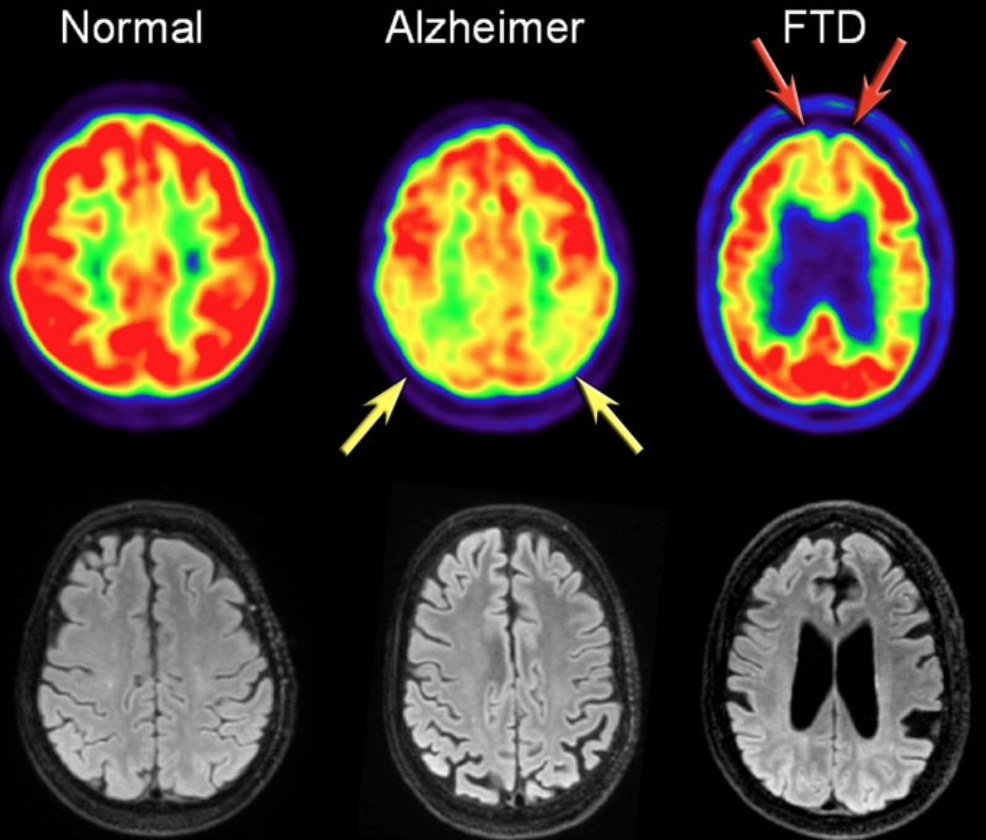
In addition to clinical findings, CSF and MRI, PET imaging are useful in diagnosing AD. In AD, FDG-PET can show hypometabolism in the temporoparietal regions and/or the posterior cingulum. This may help differentiate AD from FTD (Frontotemporal Dementia), which shows frontal hypometabolism on FDG-PET. The images show FDG-PET and axial FLAIR images of a normal subject and of patients with AD and FTD: FDG-PET (top row) and axial FLAIR images of a normal subject and of AD and FTD patients. In AD there is a decreased metabolism of the parietal lobes (yellow arrows), whereas in FTD, there is frontal hypometablism (red arrows).
TYPES OF DEMENTIA LINKED TO TBI
Alzheimer’s Disease: As mentioned earlier, Alzheimer’s disease is the most common type of dementia and is strongly associated with the buildup of amyloid plaques and tau tangles. Studies suggest that individuals with a history of TBI, especially those who experience repeated concussions, are at an increased risk of developing Alzheimer’s disease later in life.
Vascular Dementia: Vascular dementia occurs when there is reduced blood flow to the brain due to damage to blood vessels. TBI can cause damage to the brain’s blood vessels, leading to strokes or chronic ischemia (reduced blood flow) in certain regions of the brain. This vascular damage can contribute to cognitive decline and the development of vascular dementia.
Chronic Traumatic Encephalopathy (CTE): Chronic traumatic encephalopathy is a form of dementia that is specifically linked to repeated head trauma. It is most commonly found in athletes involved in contact sports, such as football or boxing and military veterans. CTE is characterized by the abnormal buildup of tau protein, and its symptoms include memory loss, mood swings, impaired judgment and cognitive decline. Individuals who experience repeated TBIs are at a significantly higher risk of developing CTE.
Frontotemporal Dementia: Frontotemporal dementia (FTD) is another type of dementia that has been associated with TBI, particularly when the injury involves the frontal and temporal lobes of the brain. FTD is characterized by changes in personality, behavior, and language, and it tends to occur earlier than other forms of dementia.
RISK FACTORS FOR DEVELOPING DEMENTIA AFTER TBI
Not everyone who experiences TBI will develop dementia, but certain factors may increase the risk. These include:
Severity of the Injury: The more severe the TBI, the higher the risk of developing dementia later in life. Severe injuries, such as those that involve loss of consciousness, long-term memory loss or extensive brain damage, are particularly risky.
Repetitive Head Injuries: Repeated TBIs, such as those sustained in contact sports or military service, significantly increase the likelihood of developing dementia. The cumulative effects of these injuries can lead to chronic traumatic encephalopathy (CTE) and other neurodegenerative diseases.
Age at the Time of Injury: Younger individuals may have a higher resilience to brain injuries, but sustaining a TBI at a young age can increase the risk of developing dementia later in life due to the long-term effects of the injury.
Genetic Predisposition: Some individuals may have a genetic predisposition to developing dementia, particularly Alzheimer’s disease. Having a family history of dementia or carrying certain genetic variants, such as the APOE ε4 allele, may increase the likelihood of developing dementia after a TBI.
Other Health Conditions: Pre-existing conditions, such as hypertension, diabetes or obesity, can increase the risk of both TBI and dementia. These conditions can exacerbate the effects of brain injury and make recovery more difficult.
PREVENTION AND TREATMENT
While there is no surefire way to prevent dementia following TBI, certain steps can be taken to reduce the risk. These include:
Protecting the Head: Using protective gear, such as helmets, during sports or activities that carry a risk of head injury can reduce the risk of TBI. Ensuring that safety measures are in place during military operations or in high-risk work environments is also essential.
Proper Management of TBI: Timely medical intervention following a TBI is critical for minimizing the long-term effects of the injury. Proper treatment can help reduce the severity of the injury and prevent complications that could increase the risk of dementia.
Avoiding Repeat Injuries: Repeated head injuries should be avoided at all costs, especially in contact sports or high-risk professions. Athletes, in particular, should be educated about the risks of concussions and the importance of following return-to-play protocols.
Healthy Lifestyle Choices: Maintaining a healthy lifestyle, including a balanced diet, regular physical activity and mental stimulation, can help protect brain health and reduce the risk of dementia. Additionally, avoiding smoking and excessive alcohol consumption can lower the risk of both TBI and dementia.
Cognitive Rehabilitation and Therapy: For individuals who have experienced a TBI, cognitive rehabilitation therapy can help improve memory, attention and other cognitive functions. Early intervention and rehabilitation can slow the progression of cognitive decline and improve quality of life.
IN CONCLUSION
The connection between TBI and dementia is increasingly recognized as a critical area of research, as the long-term effects of brain injuries become more apparent. While TBIs, especially severe and repeated injuries, increase the risk of developing dementia, understanding the mechanisms behind this connection is crucial for both prevention and treatment. By taking proactive steps to prevent head injuries and by properly managing TBI recovery, the risk of dementia can be minimized. Early diagnosis, rehabilitation and healthy lifestyle choices can further help mitigate the long-term cognitive decline associated with TBI.
The veteran reality stuntman, co-creator of the Jackass media franchise, and self-proclaimed ‘blunt force trauma guy’ may finally be rea y to stay behind the camera after suffering serious brain damage during the filming of Jackass Forever
One can’t help but make the Icarus comparison as the vision of Johnny Knoxville (aka Philip John Clapp) being shot out of a giant cannon wearing wings flashes ac oss the big screen in Jackass Forever, the ninth movie installment in this physical comedy franchise which kicked off in 2002 with Jackass: The Movie, preceded by the MTV series simply titled Jackass. As I watch Knoxville fly th ough the air in superb form, I wonder if even he thinks this may be taking things just a little too far. “When I shot out and I spread my wings like planned, I was so happy, then gravity kicked in. I started going down and turned into a big chicken in flight,” says Knoxville. “But as illie Nelson once said, ‘There’s nothing I can do about it now.’” When asked what he does for a living, Knoxville replies “I work with gravity and Newton’s third law of motion”. Well, technically, but that isn’t quite the whole picture. An American stunt performer, actor, and filmmake , Knoxville started his career in commercials and as an extra in a variety of films.
Outside of the infamous Jackass franchise, Knoxville’ filmography includes Men in Black II, A Dirty Shame, Walking Tall, The Dukes of Hazzard, and The Ringer among others. He also voiced Leonardo in Teenage Mutant Ninja Turtles. Additionally, Knoxville owns his own production company— Dickhouse Productions—and if that weren’t enough, he’s had a notable involvement with World Wrestling Entertainment, Inc. (WWE) including appearances in their Royal Rumble and SmackDown productions, facing off against Canadian professional wrestler Sami Zayn. Of all of Knoxville’s career highlights, it may seem like his involvement with the WWE could have Inspired his claimto-fame, Jackass, with their common thread of controlled, over-the-top stunt performances. However, Jackass came to be long before Knoxville stepped into the WWE ring. Some speculate that his pre-entertainment industry vocation of being a test dummy for various self-defense weapons could have been the creative spark that ignited the Jackass concept, but that wouldn’t be correct either. In fact, it was Knoxville’s failure to secure his ‘big break’ in Hollywood that prompted him to follow his natural talent for “making a spectacle of himself” [his words].
After years of chasing mainstream success in the entertainment industry, Knoxville, with help from directors Jeff remaine and Spike Jonze, pitched a television series to MTV with the simple concept of a cast of nine carrying out stunts and pranks on each other or the public, and voila, Jackass was born. The show debuted on October 1st of 2000 and the rest is history. Beginning with the television series and culminating with the most recent Jackass addition, Jackass Forever—released in February of 2022, over the years, the stunts and pranks have escalated from the benign—at least by Jackass standards—to the downright dangerous, and even PTSD inducing. “By the end of filming, they’ e [the crew] suffering f om PTSD. You can just tap them on the shoulder, and they’ll go down. They’re in terror.” says Knoxville. Take, for example, the classic escapade from the first Jackas movie, ‘Golf Course Airhorn’. This harmless—and hilarious— prank involved the Jackass crew sounding airhorns just as golfers wound up their backswings, throwing them off balance an sabotaging their endgame, the innocent antics harkening back to the simple pranks of the MTV series.
Knoxville and his crew stepped things up in later Jackass film with ‘Super Mighty Glue’, in which the crew got their hands on some extremely potent glue, and then used it to affix their bodi together and then pull them apart [ouch!], and ‘Mousetraps’ in which a member of the crew dressed as a mouse crawled through a field of mouse traps in pursuit of a piece of cheese As for Jackass stunts that Knoxville himself has performed, they have run the gamut. To name a few, there was the renta-car crash up derby, in which Knoxville came close to being crushed, the big red rocket which Knoxville rode hundreds of feet into the sky before it malfunctioned and almost blew him to pieces, and the giant evergreen tree Knoxville climbed to the top of before his crew chopped it down at its base, sending him plummeting to the ground.
The injuries that most certainly ensued after the latter mentioned stunts are undeniable, however, as the Jackass cast’s forthcoming feats proved, viewers had not seen anything yet. Enter the bulls. Bovines have held a special place in Jackass history. There was the bull that was encouraged to charge four men—one of which was Knoxville—riding a teeter-totter. And the bull whose vision was put to the test when challenged to see the camouflage Knoxville as he stood against a painted backdrop, himself painted to blend in—the bull passed the test and not only spotted Knoxville but charged at him aggressively. And finall , the less contrived stunt where a blindfolded Knoxville simply entered an enclosure with an agitated bull and just waited to be pummeled, which he was, severely. Fast forward to Jackass Forever.
Suffice it to say , Knoxville has taken things to another level, no bull, and the bodily damage it has caused is proof of that—namely a severe concussion and brain hemorrhage that caused him to lose most of his cognitive abilities for three months. In other terms, he suffe ed a traumatic brain injury (TBI). And what exactly initiated Knoxville’s TBI? You guessed it, a bull. In the most recent rendition of the Jackass staple, Knoxville enters a bullring in full magician garb and performs a magic trick for said bull, who obviously was not in the mood to be entertained. The magic show culminated in Knoxville being charged and rammed by the animal, catapulting him ten feet into the air with one and a half rotations, finally landing him squa ely on his head. “I guess that bull just didn’t like magic.” Knoxville later said. After the calamity, Knoxville lay motionless on the dirt snoring, yes snoring. “My doctor said that was me trying to swallow my tongue,” he says. It’s ironic how even his unconscious bodily response to the trauma played into Jackass’s trademark slapstick humor. All joking aside, this was serious. After about a minute, Knoxville came to, and shortly after, an ambulance shuttled him off to the hospital whe e the damage was accessed.
In the broken bones department, he got off pretty easy—just a broken rib and wrist. But in the days and weeks following the accident, the brain damage caused Knoxville to struggle cognitively and mentally. “My doctor asked me, ‘Are you having trouble concentrating?’ Apparently, I scored 17 out of 100 on a test measuring my cognitive ability. I couldn’t focus, I couldn’t edit.” Knoxville says. He also slipped into a depression—a first for him—and had debilitating headaches “My brain was just playing tricks on me. I got really depressed and over-focused on things.” Depression or no, Knoxville faced his TBI treatment headon, in true Jackass form. His team of neurologists and neuropsychologists tackled his symptoms using a combination of psychiatric medication, behavioral therapy, and transcranial magnetic stimulation (TMS), all of which are common treatments for the symptoms of TBI.
Though TBI Times is not aware of the precise medication Knoxville was prescribed, it is well-published in medical journals that selective serotonin reuptake inhibitors, commonly referred to as SSRIs, are the most effective antidep essants for people with TBI. Specificall , sertraline and citalopram—commonly known as Zoloft® and Celexa®—may have the fewest side effects an may even improve cognition. That being said, according to an article published in the Mental Health Clinician by Sophie Robert, BPharm, PharmD, BCPP, tricyclic antidepressants such as bupropion and lithium, are best avoided or used cautiously in the treatment of depressive symptoms caused by TBI.
Often prescribed as a companion treatment to psychiatric medications, cognitive behavioral therapy (CBT) is another goto for helping TBI patients experiencing emotional regulation and mental health issues—both of which CBT is extremely effective at add essing. According to a study conducted by Jennie Ponsford, Ph.D., and published in the Journal of Head Trauma and Rehabilitation, depression, anxiety, obsessive-compulsive tendencies, mood swings, impulsivity, lack of emotion, and difficulty with social interactions e all potential TBI symptoms that can be effectively t eated using CBT. Based on Michael Faraday FRS’s principle of electromagnetic induction, TMS uses low-intensity magnetic pulses to stimulate the nerve cells of the brain which some studies have shown to alleviate the mental health side effects of TBI as well as imp ove cognitive ability.
The treatment is performed in a doctor’s office while the patient is fully awake, and each session approximately 20 minutes in length. Though TMS is becoming a popular treatment for TBI, the jury is still out on its actual benefit for TBI patients. As is true with any medical treatment, results may vary, but in Knoxville’s case, his treatment combo did the proverbial trick. In his own words: “It was a really hard recovery from this last injury, but I’m great now. I feel like I’m the healthiest I’ve ever been.” Like many stuntmen and women, Knoxville has effectivel sacrificed his body for his career. He says that his doctors equate his collective trauma to “being involved in a major car crash”— aside from his TBI, the list of injuries he has incurred filmin the Jackass franchise is jaw-dropping and includes a broken collarbone, broken wrists and ribs, sprained ankles, herniated discs, torn tendons, and orbital blowout fractures. So, it isn’t surprising that after decades of extreme physical comedy and an injury list as long as his career—which spans 27 years—Knoxville is finally eady to admit defeat, or at least take a step back just shy of it. “I knew heading into this [the filming of Jackass Fo ever], that it was my last hurrah with big stunts,” says Knoxville. “You can only take so many chances before one forever catches up with you. I realized that and, amazingly, I’m still walking around. I think I’ve pushed my luck far enough.” With the next Jackass movie already in the works—Jackass 4.5— only time will tell if Knoxville stays true to his pledge to stay behind the camera
Neuroscientists in Germany and the US have recently shown that brain tsunamis, waves of cell depolarization — massive short-circuits of the neurons — sweep the cortex within ten minutes of cardiac arrest. These waves of spreading depolarization mark the beginning of the end, and trigger a gradual poisoning of neurons. They recorded brain tsunamis not just as people died but also after other critical events, such as a brain hemorrhage. Their findings coul have immediate application in emergency centers and critical-care wards.
Dr. Jens Dreier at the Center for Stroke Research Berlin and Dr. Jed Hartings at the University of Cincinnati saw an opportunity to apply these principles to their work in neurocritical care. Their centers monitor the brain activity of patients with brain conditions, such as traumatic brain injury or bleeding after an aneurysm. This neuromonitoring involves putting electrodes either directly onto the surface of the brain or deep into the cerebral cortex. Clinicians can then record electrical activity directly from the cortex.
Patients who were taken off of life-sustaining therapy whil neuromonitoring continued as the patient died revealed something striking. “Previously, it was thought that the end occurs when the brain stops its electrical activity and goes silent,” said Hartings. “But it doesn’t. We can show that the brain remains in a viable state for several minutes after this flatline, at which point a wave of depolarization sweep through the cortex. This is referred to as a brain tsunami.”
“The spreading depolarization shows that brain cells are dying, and gives a tremendously useful clinical marker for brain damage,” said Dreier. This is not just a curiosity, but something actionable in intensive care.”
By studying the brain at the end of life, these researchers have made the connection between death and spreading depolarization in a very controlled clinical setting with strong data. This may be the first step in discovering othe ways in which spreading depolarizations impact the brain and could inform breakthroughs in brain injury research and treatment.
Polytrauma is a specific medical term that describes the condition of someone who has sustained injuries to multiple body parts and organ systems. For instance, in a car crash, an individual may suffer serious burns over large portions of his or her body in addition to broken bones or a traumatic brain injury (TBI). The critical nature of an injury is evaluated in the U.S. medical community through a scale referred to as the Injury Severity Score (ISS). Polytraumas have scores of 16 or greater on this scale.
CAUSES OF POLYTRAUMA
Motor vehicle accidents are a major cause of polytrauma among civilians. The high speeds and sudden impacts that occur in many types of car crashes often lead to disastrous outcomes. The U.S. military has used the medical designation of polytrauma to categorize injuries sustained by military personnel during conflicts. Military personnel are subject to some of the most serious threats of polytrauma injuries, including blasts from explosive devices.
Beyond serious motor vehicle accidents and military-related incidents, causes of polytrauma can also include knife wounds, physical altercations, gunshot wounds, fire-related injuries, and falls from elevated heights. Polytrauma often involves a combination of TBI with other debilitating injuries such as serious eye damage, hearing damage, amputations, spinal cord injury, and severe burns. In addition, some individuals experience post-traumatic stress disorder (PTSD). Historically, polytrauma survival rates have been low, however, with advances in medical technology these statistics have greatly improved.

TREATMENT OF POLYTRAUMA PATIENTS WITH TBI
In medical terms, the presence of TBI in a polytrauma patient is referred to as Polytrauma and Concomitant Traumatic Brain Injury. According to the Journal of Neurosurgery, clinicians face unique challenges from decision-making and rehabilitative perspective when it comes to the treatment of these patients. Treatment management is complex and should be systematic, beginning at the scene with timely transport. Early operative interventions are also key.
According to the World Journal of Emergency Surgery, critical trauma care is ever-improving, yet TBI-related mortality rates are rising compared to other causes of death. Managing the acute phase after a severe TBI with polytrauma represents a challenging situation for every trauma team member and often involves the ‘damage control’ approach to sustain life. The challenge with polytrauma and concomitant traumatic brain injury patients is making sure equal emphasis is placed on stabilizing life-sustaining systems and doing everything possible to minimize brain damage.
POLYTRAUMA COULD WORSEN BRAIN INJURY
Primary brain injury results from mechanical injury at the time of the trauma whereas secondary brain injury is caused by the physiologic responses to the initial injury. Post-trauma care, as it relates to traumatic brain injury, is focused on halting or minimizing bleeding or clotting in the brain, among other biochemical processes, all of which result in secondary brain injury. Because polytrauma patients may require multiple damage control procedures, there is a risk of not providing ample emphasis on brain care. In addition, the presence of hypotension (low blood pressure), hypoxia (low oxygen levels), and fever — all commonly found in polytrauma patients — have been shown to initiate secondary brain damage.
TRAUMA NEUROSURGERY
The trauma neurosurgeon plays a key role in being able to ensure optimal treatment of polytrauma and concomitant TBI patients while minimizing secondary brain damage. Although brain surgery is highly complex and specialized, how it helps TBI patients comes down to decompression. Known as a craniotomy, this surgery relieves pressure on the brain, in turn slowing secondary brain damage. Dating back to Hippocrates, who is documented to have suggested their use in treating TBI, craniotomy revolutionized neurosurgery.
CHOOSING THE BEST TRAUMA CENTER
The best facilities for polytrauma patients to receive care are level one trauma centers. According to the American Trauma Society, A level one trauma center is capable of providing total care for every aspect of injury from prevention through rehabilitation, including surgical intervention. Although a level one trauma center can provide comprehensive treatment to trauma victims, they are not always capable of providing what is known as Simultaneous Multisystem Surgery (SMS) — which allows for different surgical teams to work on a patient simultaneously — the key to optimally treating polytrauma patients with TBI.
For a level one trauma center to provide SMS they must be equipped with a hybrid-emergency room system (HERS) where diagnostic procedures, such as CT scans, and damage control interventions, such as neurosurgery, can be performed simultaneously without patient transfer. Studies have shown that the HERS approach has been associated with a shorter time to initiate CT scanning, emergency surgery, and fewer unfavorable outcomes in polytrauma patients with and without TBI — ultimately resulting in higher – and more functional — survival rates.
While the ability to perform SMS seems to be straightforward and beneficial, very few centers in the United States are equipped with HERS and have trained staff to perform SMS in the context of TBI complicated by polytrauma. Other countries appear to be ahead of the curve in this area of trauma response. For example, not only are many of Japan’s trauma centers equipped with HERS, but they have also developed the Japanese Association for Hybrid Emergency Room Systems to specifically address the needs of specialized HERS trauma teams.
FACTORS TO CONSIDER FOR THE BEST OUTCOME
According to an article published in the National Library
of Medicine, the occurrence of TBI in polytrauma patients increases mortality and reduces their quality of life. Studies have shown that the crucial factors for ensuring the best outcomes in these cases involve getting the most appropriate care as quickly as possible. Factors such as the speed in which ambulatory care is on scene and the patient being transported to a level one trauma center — when possible one equipped to administer SMS — can make substantial differences in a patient’s recovery.
COVID-19 has been a presence in all of our lives for more than two years — and for many, so has the vaccination debate. Since the U.S. Food and Drug Administration (FDA) authorized the first COVID-19 vaccine, more than one hundred million people in the U.S. have been vaccinated. While the mainstream medical community has promoted COVID-19 vaccination — with high profile organizations like Johns Hopkins Medicine among others expressing their views that all authorized COVID-19 vaccines are highly effective at preventing serious disease, hospitalization and death from COVID-19 — there are still many who question not only the safety of COVID vaccines, but the safety of vaccines in general. A SHORT HISTORY OF VACCINES The first vaccines were based on using weaker strains of viruses to generate immunity, while not giving the recipient of the vaccine the full-blown illness or, preferably, any symptoms of the disease at all. In May 1796, a British physician named Edward Jenner tested his hypothesis that direct inoculation of a person cowpox, which is not deadly, would render that person immune to smallpox, which was. Jenner based this theory on his observations of milkmaids. Those milkmaids who had acquired cowpox through their contact with cows were immune to smallpox even when exposed multiple times to the deadly disease. Jenner’s detailed descriptions of his experiments convinced his colleagues and the authorities that inoculation with cowpox — which he called vaccination —was a viable way to protect people from smallpox. Another important figure in the history of vaccination was Louis Pasteur. Pasteur stepped onto the world stage with a famous experiment borne out of necessity. In July 1885, a rabid dog attacked a boy named Joseph Meister. At a time when this would have been a death sentence from rabies, Joseph’s mother asked for help from Louis Pasteur, who she heard was working on a cure for rabies. Pasteur inoculated the child with 13 increasingly strong doses of an experimental rabies vaccine. At the end of the treatment, the child did not develop rabies, and a new era of vaccination began.
EARLY ANTI-VACCINATIONISM
The beginnings of anti-vaccinationism were rooted in the idea of personal rights rather than health risks. In the early nineteenth century, smallpox vaccination in Europe became mandatory and societies of anti-vaccinationists formed to protest what they saw as infringement of individual liberty. Anti-vaccinationism spread to the United States later that same century, largely by visitors and immigrants from Europe, and it has been with us ever since. GENERAL VACCINE CONCERNS Vaccines, though designed to protect from disease, have been shown to cause side effects that range from mild-to-serious. According to the Centers for Disease Control and Prevention (CDC), the most common side effects of vaccination are soreness, swelling or redness at the injection site. Some vaccines

are associated with fever, rash and achiness. Serious side effects from vaccination appear to be rare, but may include lifethreatening allergic reaction, seizure and even death. However, according to the CDC, when considered on a population basis, the incidence of serious complications of vaccination is minute when compared with the outcome of natural infection. In order to understand the range of possible vaccination side effects, it is useful to compare a vaccine with relatively few associated side effects, such as the Haemophilus influenza type B vaccine, with a vaccine known to have many potential side effects, such as the smallpox vaccine, which is still used in a military context today. In the case of Haemophilus influenza type B, side effects may include redness, warmth or swelling at the injection site and elevated fever with no serious side effects having been reported. In contrast, with the smallpox vaccine, risks include encephalitis (inflammation of the brain which can lead to permanent brain damage), severe infection beginning at the vaccination site and even death — for every million people vaccinated for smallpox, up to 52 people could experience life threatening side effects.
THE CASE FOR VACCINATION
Even with anti-vaccine sentiment and vaccine concerns, the origins of vaccines do seem to make a great case for themselves, and modern science backs this up. According to the National Foundation for Infectious Diseases, The US has a robust approval process to ensure that all licensed vaccines are safe. They say that potential side effects associated with vaccines are uncommon and much less severe than the diseases they prevent. According to the CDC, getting all recommended vaccinations is a powerful step in taking charge of your health. They say that when given as directed, FDA-authorized vaccines can prevent severe disease and save lives. WHEN HARM IS CAUSED BY VACCINES When vaccines first began to be widely used, people who experienced serious side effects from vaccination had little recourse to compensation from manufacturers, physicians or the government. This was particularly a problem when vaccine production techniques were in their infancy, and contamination of vaccines occasionally occurred during or after manufacture. Since the passage in 1902 of the U.S. Biologics Control Act, which initiated the regulation of vaccines, such problems with negligence in manufacturing have greatly declined. As product liability law evolved during the 20th century, it eventually provided an avenue for compensation for individuals harmed by vaccines — they could sue vaccine manufacturers or they could sue physicians who administered vaccines. To help keep track of adverse reactions to vaccinations, the CDC and the FDA established The Vaccine Adverse Event Reporting System (VAERS) in 1990. According to the CDC, VAERS is used “to detect possible signals of adverse events associated with vaccines.” About 30,000 events are reported each year to VAERS. Between 10% and 15% of these reports describe serious medical events that lead to hospitalization, lifethreatening illness, disability or death. COVID VACCINES According to the National Foundation for Infectious Diseases, as of April 15, 2022, 19 vaccines have been authorized for emergency use and 12 given full approval for use. In 2020, the FDA authorized two mRNA COVID vaccines for emergency use. In 2021, the FDA approved the Pfizer-BioNTech COVID-19 vaccine for people ages 16 and older, and following that, the Pfizer-BioNTech vaccine for children ages 5 through 15. In 2022, the FDA approved the Moderna COVID-19 vaccine, now called Spikevax, for people age 18 and older. Researchers continue to study and develop several other COVID-19 vaccines. It’s estimated that COVID-19 vaccination results in a low risk of another infection with a similar variant for at least six months. HOW COVID VACCINES WORK According to the CDC, COVID-19 vaccines work with your immune system so your body will be ready to fight the coronavirus if you are exposed to it — including coronavirus variants. In addition, COVID-19 vaccination might offer more protection than becoming infected with COVID-19. A recent study has shown that unvaccinated people who already had COVID-19 are more than twice as likely as fully vaccinated people to be reinfected with COVID-19. Recent research also suggests that people who got COVID-19 in 2020 and then received mRNA vaccines produce very high levels of antibodies that are likely effective against current and possibly future variants. Some scientists call this hybrid immunity however further research is needed to confirm this phenomenon. DO COVID VACCINES EFFECT THE BRAIN? Though cases of neurologic symptoms related to COVID vaccination are uncommon, they are not unheard of. Two cases, published in the Cureus Journal of Medical Science, show that neurological inflammatory responses and the presence of neurooncologic lesions are possible after COVID vaccination. The journal describes two patients that presented with neurologic deficits that were not present prior to being vaccinated. One of the cases presented in the article was of a 58-year-old woman who experienced high-grade fevers, slurred speech and facial droop approximately two weeks following her second dose of a COVID-19 vaccine. According to the journal, the patient’s symptoms were attributed to post-vaccination Bell’s Palsy. Her fevers resolved over the next 24-48 hours as is typical with post-vaccination symptoms, however facial weakness persisted until approximately two weeks later when she developed acute
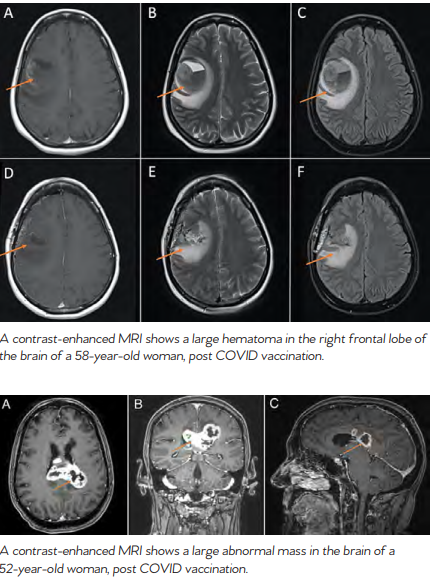
worsening of her facial droop with associated slurred speech, drooling as well as left arm and leg weakness. A contrastenhanced MRI of the brain demonstrated a large hematoma in the right frontal lobe. The patient underwent surgery to remove the hematoma. The second case presented in the journal was of a 52-yearold woman who developed severe headache and neck stiffness associated with intermittent high-grade fevers four days after her first dose of a COVID-19 vaccine. Contrast-enhanced MRI scans showed an abnormal mass on her brain. RISK VS. REWARD Despite this evidence of COVID vaccines effecting the brain, studies have shown that adverse events after COVID vaccination are rare and many COVID vaccines have proven over 90% effective with no safety threat. Additionally, other studies have shown that brain health may be more at risk after COVID infection than after vaccination. Whether or not to receive a COVID-19 vaccination is a personal decision, however, it is important to be aware of the potential adverse effects.
Each year, an estimated 1.7 million people in the United States (US) sustain a brain injury. Many treatments are being studied to help these patients to recover and regain their normal lives — art therapy is one. Art therapy is a distinct discipline that incorporates creative methods of expression through visual art media. Art therapy, as a creative arts therapy profession, originated in the fields of art and psychotherapy and may vary i definition. This modality aids in the ecovery process by allowing the patient to do something they enjoy while working on various functional skills such as fine moto skills, gross motor skills, standing tolerance, endurance, communication, expression of feelings, relaxation, socialization, memory, and problem-solving skills. In the inpatient setting, art therapy can be used daily with both pediatric and adult patients. This form of treatment allows patients to be creative while simultaneously motivating them. The format for this type of therapy can be straightforward, for example, drawing and painting, or the approach can be much more creative. Take for example a pediatric patient with traumatic brain injury who was very interested in coloring and painting. To help with her walking and coordination, her therapist painted her feet and had her walk throughout the facility. This allowed the patient and her family to see the progress she made through the footprints she created. She was allowed to express herself daily by choosing the colors for her footprints and was motivated to continue walking more each day.

Art therapy is also being explored as a form of complementary and integrative care for military veterans affected by trauma and injuries in the line of duty includin traumatic brain injury (TBI) and post-traumatic stress disorder (PTSD). The self-soothing qualities of making art can most certainly aid in TBI recovery. Art therapy offers many psychologica and cognitive benefits for patients after a TBI and als helps improve mood, problem-solving skills, attention, and coordination. Most importantly, making art helps patients find a healthy outlet for their emotions, and ebuild their sense of self, something few other therapies can offer after traumatic brain injury.
According to John Hopkins Medicine, Body Dysmorphic Disorder (BDD) is a mental illness that causes a person to be obsessively focused on a perceived flaw in their appearance. A person with BDD may be so preoccupied with the appearance of their body that they cannot lead a normal life and may be overcome with feelings of self-hate and dissatisfaction. They may spend an excessive amount of time each day worrying about how they look, so much so that they neglect their daily responsibilities. Suicidal thoughts may also be a symptom. Some behaviors that may accompany this disorder include a person constantly checking themselves in the mirror, avoiding social activities have shown that individuals with BDD have deficiencies in this area. In one study of verbal and nonverbal memory — including visual organization strategies — subjects were asked to copy and recall a complicated figu e drawing.
The BDD group recalled more specific parts of the drawin instead of the overall structure, which may reflect poo organizational strategies marked by an imbalance in local (detail) and global information processing. Another study using the same test found impaired copying and recall in individuals with BDD. There was also evidence of impaired visual working memory as well as auditory, verbal and logical memory.
“THERE IS NO QUESTION THAT PHYSICAL DAMAGE TO THE BRAIN CAN CAUSE
COGNITIVE, EMOTIONAL, AND BEHAVIORAL ISSUES. RESEARCH HAS SHOWN THAT
THE SPECIFIC AREA OF THE BRAIN AFFECTED BY TBI TRANSLATES DIRECTLY TO THE
POTENTIAL SIDE EFFECTS A PATIENT MAY EXPERIENCE.”
- Dr. Rod Amiri, MD, Neuropsychiatrist
New research has started to identify abnormal areas and connections in the brain in hopes of developing biomarkers or brain correlates that can help identify those at risk for developing BDD. Medical research is beginning to make connections between the disorder and frontal lobe brain damage. Trauma or injury to the frontal lobe of the brain can cause a wide range of problems and changes to your personality due to the frontal lobe’s role in shaping social behavior and personal characteristics. It controls things such as personality, decision making, motivation and voluntary movements. The frontal lobe is also responsible for memory, and studies. Individuals with BDD also tend to do poorly in tasks related to decision-making, specifically those involving planning, inhibition or organization, pointing again to the frontal lobe. One study found that compared to healthy controls, those with BDD made more errors on a search task, demonstrating deficits in working memor . They also were slower on a task measuring planning ability and exhibited higher risk-taking behavior in a decision-making experiment. Based on these studies, the connection between frontal lobe abnormality and BDD is clear. However, because of the complexity of the disorder, future research is necessary to understand the exact combiniation of factors that lead to BDD.
War ravages lives across borders and cultures and within military ranks. The physical casualties [of war] are front and center, but the less talked about and equally debilitating occurrences of traumatic brain injury (TBI) are the silent wounds effecting millions globally — wounds which often goes untreated. In a conflict setting, TBI is commonly caused by blasts and leaves service members and civilians alike to grapple with life-altering cognitive difficulties, sometimes without adequate medical care to support recovery. Though the problem is more prevalent than the help, there are many agencies and organizations dedicated to assisting these victims of war.
The Office of the UN High Commissioner for Human Rights (OHCHR), the UN Assistance Mission (UNAMA), the United Nations Children’s Fund (UNICEF), and others, are dedicated to shedding light on the problem and providing solutions. SERVICE MEMBERS AND TBI According to the Joint Theater Trauma Registry, compiled by the U.S. Army Institute of Surgical Research, 22 percent of soldiers wounded in the U.S. war in Afghanistan had injuries to the head, face or neck. This percentage can serve as a rough estimate of the fraction who have TBI, according to Deborah L. Warden, a neurologist and psychiatrist at Walter Reed Army Medical Center who is the national director of the Defense and Veterans Brain Injury Center (DVBIC). Warden said the true proportion is probably higher, since some cases of closed brain injury are not diagnosed promptly.
In the Vietnam War, by contrast, 12 to 14 percent of all combat casualties had a brain injury, and an additional 2 to 4 percent had a brain injury plus a lethal wound to the chest or abdomen, according to Ronald Bellamy, former editor of the Textbooks of Military Medicine, published by the Office of the Surgeon General of the U.S. Army. Bellamy said that because mortality from brain injuries among U.S. combatants in Vietnam was 75 percent or greater, soldiers with brain injuries made up only a small fraction of the casualties treated in hospitals. Kevlar body armor and helmets are one reason for the high proportion of TBIs among soldiers wounded in more current conflicts. They work by shielding the wearer from bullets and shrapnel and have improved overall survival rates.
Kevlarhelmets have reduced the frequency of penetrating head injuries. However, the helmets cannot completely protect the face, head and neck — nor do they prevent the kind of closed brain injuries often produced by blasts. Most brain injuries in war zones are caused by improvised explosive devices (IEDs), and closed brain injuries outnumber penetrating ones among patients seen at Walter Reed, where thousands of patients with TBI are treated. All admitted patients who have been exposed to a blast are routinely evaluated for brain injury — 59 percent of them have been given a diagnosis of TBI, according to Warden.
Of these injuries, 56 percent are considered moderate or severe, and 44 percent are mild. A blast creates a sudden increase in air pressure by heating and accelerating air molecules and, immediately thereafter, a sudden decrease in pressure that produces intense wind. These rapid pressure shifts can injure the brain directly, producing concussion or contusion. Air emboli — a blockage of blood supply caused by air bubbles — can also travel to the brain, causing the death of brain tissue. When service members have serious brain injuries, they receive immediate care on the battlefield and are then transported to military combat support hospitals, where they undergo brain imaging and are treated by neurosurgeons.
Treatment may include the removal of foreign bodies, control of bleeding or craniectomy — which relieves pressure from swelling. Soldiers with TBI often have symptoms affecting several areas of brain function. Headaches, sleep disturbances and sensitivity to light and noise are common. Cognitive changes, diagnosed on mental-status examination or through neuropsychological testing, may include disturbances in attention, memory or language, as well as delayed reaction time during problem solving. Often, the most troubling symptoms are behavioral ones — mood changes, depression, anxiety, impulsiveness, emotional outbursts or inappropriate laughter.

Some symptoms of TBI overlap with those of post-traumatic stress disorder, and many effected in the military sector have both conditions. The good news is service men and women are young and healthy, and as such are in a good position to recover. CIVILIANS INJURED IN CONFLICTS The U.S. wars in Iraq, Afghanistan, Yemen, Syria and Pakistan, military operations in Somalia as well as the more recent conflict in Ukraine have taken a tremendous human toll on those countries. Hundreds of thousands of civilians have been injured as a result of these conflicts and many of these injuries have resulted in TBI. For example, according to the Watson Institute for International and Public Affairs, blast-induced TBI has been referred to as the signature injury of the conflicts in Iraq and Afghanistan. People living in these war zones have been attacked in their homes, in markets and on roadways. They have been subjected to bombs, bullets, fire and IEDs.
Civilians are victimized at checkpoints, as they are run off the road by military vehicles, when they step on mines or cluster bombs, as they collect wood or tend to their fields and when they are kidnapped and executed for purposes of revenge or intimidation. A new book, published by world-renowned UCSF neuroradiologist Dr. Alisa Gean, tackles the complexity of TBI, how it is sustained and how it affects both civilians and combatants alike. The text includes information and research gathered from nearly three decades of studying civilian TBI, as well as five intensive years of studying TBI sustained from combat, terrorism and natural disasters. “Aproximately 85% of injured World War II soldiers suffered from gunshot wounds.
Today’s enemy is using a new weapon to kill, defeat and demoralize — the improvised explosive device.” says Dr. Gean. The U.S. war in Afghanistan lasted for 22 years and resulted in an enormous footprint of civilian casualties. In their Afghanistan Protection of Civilians in Armed Conflict Annual Report, the Office of the UN High Commissioner for Human Rights (OHCHR) and the UN Assistance Mission (UNAMA) documented some 8,820 civilian casualties (3,035 deaths and 5,785 injuries) in 2020. These civilians paid a terrible price for the failure of peace negotiations and the country remains amongst the “deadliest places in the world to be a civilian”, according to Michelle Bachelet, UN High Commissioner for Human Rights. “I am particularly appalled by the high numbers of human rights defenders, journalists and media workers killed since peace negotiations began”, she says.
According to the report, the overall drop in civilian casualties in 2020 was due to fewer casualties from suicide attacks by anti-government elements in populated areas, as well as a drop in casualties attributed to international military forces. There was, however, a “worrying rise” in targeted killings by such elements — up about 45 per cent since 2019. The use of IEDs by the Taliban, air strikes by the Afghan Air
Force and ground engagements also resulted in increased casualties, according to the report. Anti-government elements bore responsibility for about 62 percent of civilian casualties, while pro-government forces were responsible for about 25 pe cent of the casualties. About 13 percent of casualties were attributed to crossfire and other incidents. The report went on to note that the years-long conflict in Afghanistan “continues to wreak a shocking and detrimental toll” on women and children, who accounted for 43 percent of all civilian casualties — 30 percent children and 13 percent women. “This report shows the acute and lasting needs of victims of the armed conflict and demonstrates how much remains to be done to meet those needs in a meaningful way”, High Commissioner Bachelet said. “The violence that has brought so much pain and suffering to the Afghan population for decades must stop and steps towards reaching a lasting peace must continue.”

June 3rd marked the 100th day of the war in Ukraine — a war that has shattered the lives of millions of children. Only days before, on June 1st, the International Day for Protection of Children [in Ukraine and across the region] was marked. “June 1 is the International Day for Protection of Children,” said UNICEF Executive Director Catherine Russell. “Instead of celebrating the occasion, we are solemnly approaching the 100th day of the war. Without an urgent ceasefire and negotiated peace, children will continue to suffer — and fallout from the war will continue to impact vulnerable children around the world.” The scale and speed of the emergency in Ukraine have not been seen since World War II, the United Nations said in a statement, which estimated that 5.2 million children in the region need humanitarian assistance; three million inside the country and more than 2.2 million in refugee-hosting countries. As intense artillery exchanges continue between Russian and Ukrainian forces in eastern Donetsk region and amid reports that Russian troops are continuing their campaign, UNICEF said that almost two in three children in Ukraine have been displaced by fighting. “Civilian infrastructure on which children depend continues to be damaged or destroyed,” the agency explained. “This so far includes at least 256 health facilities and one in six UNICEF-supported ‘Safe Schools’ in the country’s east. Hundreds of other schools across the country have also been damaged.
Conditions for children in eastern and southern Ukraine where fighting had been quite intense are increasingly desperate.” In addition to the trauma of fleeing their homes, UNICEF states that children fleeing violence faced a significant risk of family separation, abuse, sexual exploitation and trafficking. The UN agency also reiterated its call for “full humanitarian access” so that teams can “safely and quickly reach children in need wherever they may be”. Inside Ukraine, UNICEF and partners have already distributed life-saving health and medical supplies for nearly 2.1 million people in war-affected areas. Equally important, critical safe water access has also been secured for more than 2.1 million people living in areas where networks have been damaged or destroyed.
The long-lasting mental health toll of the war on children has also contributed to an acute child protection crisis, made worse by the fact that many displaced families are out of work and unable to meet their children’s basic needs. “These children urgently need safety, stability, child protection services and psychosocial support — especially those who are unaccompanied or have been separated from their families. More than anything, they need peace,” UNICEF insisted. Over 610,000 children and caregivers have also received mental health and psychosocial support, while nearly 290,000 children have been given learning supplies. In addition, almost 300,000 vulnerable families have registered for a humanitarian cash assistance program run by UNICEF and the Ukrainian Ministry of Social Policy. In countries hosting Ukrainian refugees, the UN agency continues to look out for the most vulnerable of children by providing anti-trafficking training for border guards and encouraging local authorities to integrate refugee children into schools.
Providing vaccines and medical supplies for displaced Ukrainians is a key component of UNICEF’s response, and so too is establishing play and learning hubs to provide young children with a much-needed sense of normalcy and respite. In total, 25 UNICEF-UNHCR “Blue Dots” — one-stop safe havens that provide support and services for families on the move, have been established along major transit routes in Moldova, Romania, Poland, Italy, Bulgaria and Slovakia, the agency said.
In Moldova, more than 52,000 refugees, mostly in female-headed households, have been reached through a UNICEF-UNHCR multi-purpose cash assistance program. Highlighting the devastating and lasting consequences of the war on millions of the country’s youngest citizens, UN Children’s Fund UNICEF maintains that most of the victims were from “attacks using explosive weapons in populated areas”, a claim 1.6 BILLION CHILDREN ARE LIVING IN A CONFLICT AFFECTED COUNTRY supported by the UN human rights office, OHCHR. With the use of explosive weapons, it is inevitable that many of these children will be effected by TBI. According to the Save the Children Blast Injury Report, the use of explosive weapons, traditionally meant for the open battlefield, in densely populated towns and cities continues to cause devastation to children.
Time after time, these weapons result in death, life-changing injuries and the destruction of vital facilities, such as schools and hospitals. From Syria, to Yemen, to Afghanistan, the families of the injured are left to manage TBI among other devastating injuries with health systems often at the point of collapse. And children are particularly vulnerable to blasts with their bodies being lighter, and thus easily thrown farther than an adult’s body. There are good reasons why the killing and maiming of children in conflict is defined as a grave violation by the United Nations.
If children survive explosive weapons, they often find themselves dealing not only with physical trauma and disability, but with the loss of family members, the destruction of their homes and the disruption of their education — and with it their future prospects. To say nothing of the acute stress already caused by growing up in a war zone. Though children can be quite resilient, they cannot be expected to recover without sustained and specialist support. Left untreated, the long-term effects of blast injuries will leave children facing a lifetime of suffering, and will ultimately create an entire generation at risk of exclusion. The Pediatric Blast Injury Partnership (PBIP), led by Imperial College London with Save the Children, is a practical response to the unique medical challenges faced by children with blast injuries. One of the PBIP’s most valuable resources to date is the Pediatric Blast Injury Field Manual, which gives medical staff in conflict settings, who often have to operate with little or no previous experience or training, the knowledge and technical guidance needed to treat children from the point of injury onwards. It also offers advice on how to provide long-term rehabilitation care and mental health support.
Smart initiatives like these will make a real difference to injured children, and medical institutions and humanitarian agencies like Save the Children will undoubtedly continue to build on this work to ensure children have the best chance of recovery. But in order to truly protect children in conflict, governments need to take an active role to advocate for children. This is why PBIP is calling on all warring parties to Stop the War on Children by taking steps to uphold international laws and norms, ensuring accountability for crimes against children and investing in the support necessary to help children recover from the physical and psychological trauma caused by conflict. PBIP found that 80% of pediatric blast patients experienced penetrating injuries to the head. By comparison, just 31% of adult blast patients experienced the same injury. Within this, younger children are more affected. Children under seven are almost twice as likely to present with head injuries as older children. In one study, 90% of children who died from blast injuries had skull fractures.
When children survive a blast, they are almost always left with life-limiting brain injury. Take 12-year-old Mahmoud who lives in Gaza. In 2014 when he was playing in the street, he was hit by an explosive weapon. “I heard an explosion and I felt something go into my eye. I touched my eye and felt blood pouring out. I ran for help and was taken to the hospital. I woke up missing one of my eyes.” Mahmoud’s family says byond his physical injuries, he was not himself after the accident. Mahmoud ikely suffered an undiagnosed TBI. Helle Thorning-Schmidt, CEO of Save the Children International and former Prime Minister of Denmark is uncompromising in her assessment: “International law makes clear that everyone has a responsibility to make sure children are protected in war. Yet explosive weapons continue to kill, maim and terrorize thousands of children every year. Every warring party — from armed groups to governments — must do more to protect children and abide by this important moral principle.”
Contribute to the TBI Times






