Traumatic brain injury (TBI) — caused by blows, impacts, falls, or blast exposure — remains a major source of long-term disability worldwide. Many survivors struggle with lingering problems in memory, movement, mood regulation, and sensory processing. Traditional treatments can help manage symptoms but do little to actively repair damaged neural circuits. In recent years, a breakthrough technology known as optogenetics has emerged as a powerful new way to study and potentially treat the underlying disruptions caused by TBI.
What Is Optogenetics?
Optogenetics is a technique that uses light to control specific neurons in the brain. Scientists introduce light-sensitive proteins, called opsins, into targeted brain cells using harmless viral vectors. Once these proteins are expressed, shining a precise wavelength of light on the cells can turn them “on” or “off.” This allows researchers to control neural activity with an unprecedented level of precision — not just on the scale of specific brain regions, but down to individual types of neurons and millisecond-timing.
This level of accuracy sets optogenetics apart from traditional electrical stimulation, which affects all nearby cells indiscriminately. Instead, optogenetics allows researchers to target exactly the circuits they want to study or modulate, making it ideal for decoding the complex changes caused by traumatic brain injury.
How Optogenetics Helps Us Understand TBI
Researchers have begun using optogenetics in animal models to explore how the brain rewires and functions after injury. These studies are revealing key insights:
- Tracking how neurons change after injury: By stimulating specific neurons and observing their responses, scientists can map how TBI alters neural firing patterns and communication. This has shown that many neurons become less responsive or misaligned with normal brain rhythms, helping explain long-term cognitive and motor impairments.
- Probing circuit rewiring: After TBI, damaged circuits often attempt to reorganize. Optogenetics enables researchers to stimulate precise pathways and see how other regions compensate over time. For example, the hemisphere opposite the injury sometimes takes on greater control of movement, showing the brain’s natural drive to adapt and recover.
- Improving memory and cognition in models: In several studies, activating specific populations of neurons after injury improved tasks related to learning and memory. Stimulating newborn neurons in the hippocampus — a region essential for memory — helped them survive and integrate better, leading to improved cognitive performance.
- Studying blood flow and metabolic changes: TBI often disrupts neurovascular coupling, the mechanism that links neural activity to blood flow. Using light to activate neurons, researchers can study how blood vessels respond and identify strategies to restore healthy circulation in injured tissue.
Toward Therapeutic Use
While most work remains in the experimental stage, the therapeutic potential of optogenetics is growing. Possible future strategies include stimulating surviving neurons to enhance recovery, guiding the brain’s natural plasticity to rebuild circuits, or improving blood flow in damaged areas.
However, several challenges remain before optogenetics can be used clinically. Delivering opsins safely to human neurons is complex, and reliably delivering light deep into the brain without invasive equipment is still a major engineering hurdle. Additionally, long-term safety studies are needed.
Looking Ahead
Optogenetics is transforming our understanding of how the brain responds to traumatic injury. Though not yet ready for clinical use, it offers a powerful new window into TBI and provides hope for therapies that go beyond symptom management to truly repair damaged circuits. As research progresses, this light-based approach may one day play a critical role in helping TBI survivors regain lost function and improve quality of life.
Traumatic brain injuries (TBIs) are among the most time-sensitive and challenging emergencies first responders face. Whether caused by vehicle collisions, falls, sports incidents, or assaults, a TBI can rapidly worsen without proper early intervention. As the frontline professionals who assess and stabilize patients in the critical first minutes after injury, first responders play a pivotal role in determining outcomes. That’s why specialized TBI training is becoming an essential part of emergency medical practice.
For decades, TBI was often treated as a secondary concern during trauma response. Visible injuries—bleeding, broken bones, airway obstruction—drew attention first. But research and experience have shown that early recognition of a brain injury can dramatically affect survival rates and long-term recovery. Modern first responder training emphasizes that a patient who “looks fine” can still be at risk for life-threatening intracranial damage.
A core component of TBI-focused training is recognition of subtle symptoms. First responders are taught to look beyond obvious signs like loss of consciousness. Confusion, delayed responses, nausea, slurred speech, unusual behavior, or even a headache may signal a serious brain injury. Because adrenaline can mask symptoms, and because children and older adults may present atypically, responders are trained to assume a TBI until proven otherwise in many high-risk scenarios.
Another vital element is managing the airway and preventing secondary brain injury. Hypoxia and hypotension can significantly worsen outcomes for TBI patients. Updated training protocols emphasize maintaining adequate oxygenation and blood pressure while avoiding unnecessary movement of the head and neck. Spinal precautions remain essential, especially when mechanism of injury suggests possible concurrent trauma. First responders also practice controlled ventilation techniques, as both under- and over-ventilation can harm a brain struggling to regulate pressure.
Modern training also incorporates advances in field assessment tools. Many departments now use standardized tools such as the Glasgow Coma Scale, pupil reactivity testing, and concussion screening checklists. Some regions are piloting portable brain imaging devices or infrared spectroscopy tools to detect intracranial bleeding in the field. While not yet universally available, familiarity with these technologies ensures first responders can integrate them seamlessly into practice as they become standard.
Equally important is communication and documentation. The way first responders describe the patient’s initial condition—mental status, mechanism of injury, any changes during transport—helps emergency departments determine next steps quickly. A drop in GCS or a report of vomiting or disorientation, even if brief, can influence decisions such as whether to perform CT scanning or consult neurosurgery immediately.
Finally, TBI training now emphasizes the human side of emergency response. First responders must know how to reassure frightened patients, keep them still and calm, and communicate clearly with family members who may not understand the severity of what’s happening. Behavioral symptoms like agitation or emotional instability are common in TBI patients and require skilled, compassionate handling.
As our understanding of brain injuries expands, first responder training continues to evolve. By equipping emergency personnel with the knowledge and tools to identify TBIs early, protect vulnerable patients, and prevent complications, we improve not only survival rates but long-term quality of life for thousands of people each year.
Disclaimer: This article is for informational purposes only and is not medical or legal advice.
Traumatic brain injury (TBI) often leads to significant disruptions in sleep, a crucial component of physical and cognitive recovery. Sleep disturbances are common after a TBI, and these disruptions can worsen other symptoms such as memory problems, mood swings and increased fatigue, further complicating the recovery process. Understanding the relationship between TBI and sleep is essential for improving patient outcomes and helping individuals regain their quality of life.
After a TBI, sleep problems can manifest in various ways, including difficulty falling asleep, frequent waking during the night or excessive daytime sleepiness. These disturbances are linked to the brain’s inability to properly regulate sleep-wake cycles, often due to damage in areas that control sleep, such as the hypothalamus. Additionally, the emotional and physical trauma from the injury can contribute to stress, anxiety and depression, all of which can further disrupt sleep patterns. Post-concussion syndrome, which is common after TBI, can result in prolonged sleep issues, with many patients struggling with insomnia or disturbed sleep for months or even years.
To address these sleep disturbances, a multifaceted approach is often necessary. Cognitive behavioral therapy for insomnia (CBT-I) has shown to be an effective, non-pharmacological treatment, helping individuals alter behaviors and thought patterns that interfere with sleep. It focuses on improving sleep hygiene, relaxation techniques and the development of a consistent sleep schedule. Medications may also be used to manage specific sleep issues, such as sleep aids to help initiate sleep or antidepressants to address mood disorders that affect sleep. However, medications should be carefully prescribed and monitored to avoid dependency or adverse effects that could interfere with brain healing.
Lifestyle adjustments can also play a role in improving sleep quality. Establishing a calming bedtime routine, limiting screen time before bed and creating a quiet, comfortable sleep environment can promote better rest. By addressing the effects of TBI on sleep through both behavioral and medical interventions, individuals can improve their recovery process, reduce fatigue and enhance overall well-being.
Genetic research is rapidly advancing the field of traumatic brain injury (TBI) by providing insights into how individuals respond to brain injuries and the potential for personalized treatments. TBI, which results from external forces damaging the brain, can lead to a wide range of cognitive, emotional and physical impairments. While some individuals recover with minimal long-term effects, others may experience severe, lifelong consequences. Genetic research is helping to explain why these outcomes vary and offers the potential to improve diagnosis, treatment and prevention.
One key area of genetic research in TBI involves identifying genetic variations that influence an individual’s susceptibility to injury and their ability to recover. Studies have shown that certain genes, particularly those involved in inflammation, cellular repair and neuronal survival, can play a significant role in the severity of injury and the brain’s capacity to heal. For example, variations in the apolipoprotein E (APOE) gene, known for its link to Alzheimer’s disease, have been shown to affect how the brain responds to TBI. Some variations of the APOE gene increase the risk of cognitive decline following a TBI, while others may offer a degree of protection.
Another promising area of research focuses on genes related to oxidative stress and inflammation, both of which are activated following a brain injury. These processes can cause further damage to brain cells and contribute to the development of conditions such as post-traumatic stress disorder (PTSD) or neurodegenerative diseases. By understanding how these genetic factors influence the brain’s response, researchers are working toward identifying individuals who may be at higher risk for these long-term complications.
Additionally, genetic studies are aiding in the development of more targeted treatments. With knowledge of an individual’s genetic makeup, doctors may be able to recommend personalized therapies that improve recovery outcomes. For instance, genetic testing may help identify patients who could benefit from certain pharmaceutical treatments or rehabilitation strategies, improving the effectiveness of interventions.
In conclusion, genetic research is advancing TBI research by revealing the underlying biological factors that influence injury severity and recovery. These discoveries are leading to more personalized, effective treatments and offering hope for better outcomes for individuals with TBI.
Transcranial magnetic stimulation (TMS) is gaining attention as a noninvasive treatment for traumatic brain injury (TBI), offering a novel approach to help patients recover from the cognitive, emotional and physical impairments caused by brain trauma. TBI, which results from an external force damaging the brain, often leads to long-lasting symptoms such as memory problems, difficulty with attention, mood disorders and motor dysfunction. Traditional treatments focus on rehabilitation and symptom management, but TMS offers the potential to directly influence brain activity and facilitate recovery.
TMS works by using magnetic fields to generate electrical currents in specific areas of the brain. During treatment, a magnetic coil is placed on the scalp, producing a magnetic pulse that stimulates nerve cells in the targeted region of the brain. The stimulation can increase or decrease neural activity, depending on the parameters used, helping to restore normal brain function in areas affected by the injury. TMS has been shown to improve brain plasticity, which is the brain’s ability to reorganize and form new connections.
Research into TMS as a treatment for TBI is still in its early stages, but preliminary studies have shown promising results. TMS has been found to improve cognitive functions such as attention, memory and executive functions, which are often impaired after a brain injury. Additionally, TMS may have a positive effect on mood and emotional regulation, helping to alleviate symptoms of depression and anxiety that are common in TBI patients.
While TMS offers a noninvasive and relatively safe treatment option, challenges remain in optimizing the treatment protocols for TBI patients. These include determining the most effective stimulation parameters, identifying the best candidates for treatment and understanding the long-term effects of TMS. However, as research continues to explore its potential, TMS holds promise as a valuable tool in the rehabilitation and recovery process for individuals with traumatic brain injury, offering hope for better functional outcomes and improved quality of life.
Neurotrophins are a group of proteins that play a critical role in the growth, survival and repair of neurons in the brain and nervous system. These molecules are vital for maintaining the health and function of neurons, the cells responsible for transmitting signals throughout the body. Following brain injuries, such as traumatic brain injury (TBI) or neurodegenerative diseases, neurotrophins help facilitate the repair and regeneration of damaged brain cells, promoting recovery and improving cognitive and emotional function.
One of the primary neurotrophins is brain-derived neurotrophic factor (BDNF), which supports the survival and growth of neurons in the brain. BDNF plays an essential role in neuroplasticity, the brain’s ability to reorganize and form new connections. This is particularly important after a brain injury when the brain must repair damaged neural pathways and form new ones to restore function. By promoting the growth of new synapses and enhancing communication between neurons, BDNF aids in the recovery of memory, learning and motor functions often impaired after injury.
Another key neurotrophin is nerve growth factor (NGF), which is important for the survival and maintenance of certain types of neurons, particularly those involved in the autonomic nervous system. NGF is involved in the regeneration of nerve fibers after injury, and its action helps restore function to damaged neural networks. Additionally, it has been shown to reduce cell death in response to brain injury, further supporting the repair process.
Neurotrophins can also reduce the harmful effects of inflammation in the brain. After a TBI, inflammation can cause further damage to brain cells and delay recovery. By regulating inflammatory responses and promoting neuroprotection, neurotrophins can help limit secondary damage and enhance the brain’s ability to heal.
Researchers are investigating various therapies that can stimulate neurotrophin production or mimic their effects, such as using drugs or gene therapies to increase levels of these proteins in the brain. These approaches hold promise for improving the recovery of brain function after injury or in conditions like Alzheimer’s disease or Parkinson’s disease. Ultimately, neurotrophins play a vital role in the brain’s ability to repair and regenerate itself, offering hope for enhancing recovery and restoring cognitive health after brain injury.
Color therapy, also known as chromotherapy, is a form of alternative treatment that uses colors to influence mood, physical health and emotional well-being. In the context of traumatic brain injury (TBI), color therapy has gained attention as a potential complementary treatment to help alleviate symptoms such as anxiety, depression and cognitive difficulties. While it is not a standalone treatment for TBI, research suggests that it may have therapeutic benefits by stimulating specific brain areas and promoting a sense of balance and relaxation.
Colors have been shown to have different effects on the human body and mind. For example, the color blue is known for its calming properties and is often used to reduce stress and anxiety. After a TBI, individuals may experience heightened anxiety, agitation or sleep disturbances, and the soothing effects of blue may help promote relaxation and better sleep. Similarly, the color green, associated with balance and harmony, is often used to reduce emotional stress and increase mental clarity. For TBI patients experiencing mood swings or cognitive difficulties, green can be used to create a calming environment that aids in concentration and emotional stability.
Red, on the other hand, is a color associated with energy and stimulation. While it may not be suitable for all TBI patients, red can be used strategically to stimulate brain activity and encourage mental alertness. This can be particularly helpful for those dealing with fatigue or cognitive slowness, as red may help enhance focus and motivation.
Yellow, a color linked to positivity and optimism, is also used in color therapy to stimulate mental activity and improve mood. For TBI patients who struggle with depression or a sense of hopelessness, exposure to yellow can help lift spirits and promote a more optimistic outlook on recovery.
While color therapy is not a replacement for medical treatments, it can be a valuable adjunct to traditional therapies, helping TBI patients manage symptoms and improve overall well-being. By incorporating color therapy into rehabilitation programs, individuals may experience greater emotional balance, mental clarity and a sense of calm during their recovery journey.
Fasting has gained attention in recent years for its potential health benefits, and research suggests that it may help alleviate symptoms associated with traumatic brain injury (TBI). TBI, which can result from blows or jolts to the head, often leads to cognitive, emotional and physical impairments. While traditional treatments primarily focus on rehabilitation and symptom management, fasting presents a novel approach that could support recovery by enhancing brain function and promoting healing.
Fasting triggers several biological processes that may benefit TBI patients. One of the key mechanisms is the activation of autophagy, a process where the body cleans out damaged cells and regenerates healthier ones. This process is essential for repairing brain tissue and supporting neuronal health. After a TBI, the brain may experience inflammation and cell damage, and autophagy can help mitigate these effects by clearing damaged cells and promoting neurogenesis, the growth of new neurons.
Additionally, fasting can enhance the production of brain-derived neurotrophic factor (BDNF), a protein that supports the survival of existing neurons and encourages the growth of new ones. BDNF plays a crucial role in neuroplasticity, which allows the brain to reorganize and adapt following an injury. Increased levels of BDNF could help improve cognitive function, memory and emotional regulation, which are often impaired after a TBI.
Fasting may also help reduce inflammation, which is a common issue after brain injury. Inflammation can exacerbate brain damage and contribute to long-term cognitive deficits. By reducing inflammation, fasting may aid in minimizing further brain injury and improving overall recovery.
While more research is needed to fully understand the impact of fasting on TBI recovery, early findings suggest that it could be a useful adjunct to traditional therapies. It is essential, however, for individuals considering fasting to consult with healthcare providers, as it may not be suitable for everyone, especially those with underlying health conditions. Nonetheless, fasting offers a promising area of exploration for improving TBI symptoms and supporting brain healing.
Every year, 1.7 million people in the US sustain a traumatic brain injury (TBI). Psychiatric disorders frequently complicate the course of recovery from TBI and occur at rates exceeding those of the general population. Major depression is the most common psychiatric disorder following TBI, affecting an estimated 29.4% of patients in the first year post-injury alone. TBI-associated depression contributes to higher suicide risk, altered executive function, poorer social reintegration, and vocational outcomes, and decreased quality of life.
TBI has long been associated with changes in mood, personality, and behavior. Research suggests that psychiatric disorders may be present at increased rates after TBI and for this reason, TBI is considered by some medical professionals to be a risk factor for psychiatric diagnosis. “One example of how TBI can be connected with mood or anxiety disorders is when the cognitive complications associated with the TBI result in profound changes in the patient’s life. If they can no longer work and experience financial hardship or loss in purpose, this can lead to depression and anxiety,” says Dr. Emin Gharibian, Psy.D., founder, and director of Verdugo Psychological Associates.
Research has shown that the specific area of the brain affected by the TBI translates directly to the potential side effects a patient may experience. For example, injuries on the right side of the head are more likely to cause manic symptoms, and injuries on the left side of the head are more likely to cause depressive symptoms. Frontal-cortex injuries can lead to behaviors that are typically associated with personality disorders. This is because the frontal-cortex region of the brain is directly responsible for emotional regulation.
The chronic and relapsing course of TBI-associated depression poses a challenge to the management of afflicted patients. Two-thirds of patients depressed at 1-year post-injury remain so in the second year, and the risk of depression remains elevated for 20 to 30 years after the injury. The high prevalence, chronicity, and potentially irreversible consequences of post-TBI depression underscore the importance of developing interventions targeting this disorder.

The length of time it takes for a patient with TBI to recover directly influences their chances of facing long term depression. With an extended recovery time, a patient’s socialization, work, autonomy, or even their ability to communicate will be affected. Changes in your typical routines and habits have a direct effect on your mental state and quality of life. Because of that, lingering TBI symptoms are strong activators of the behavioral inhibition system, causing symptoms of depression. Once the behavioral inhibition system is disturbed, the ability to feel pleasure or satisfaction can be lost.
For TBI patients, not only is that inhibition system overactive, but they’ve lost many of the activities that could jumpstart their behavioral activation system (which would help calm down the inhibition system and allow feelings of reward and pleasure again). Other factors to consider are sleep and pain. Many TBI patients experience disruption to their normal sleep patterns, and sleep deprivation itself can result in depressive symptoms. Additionally, the symptoms of depression can make it harder to sleep, so they work together to make everything feel worse. Coping with continuous pain is another factor. It is difficult for the brain chemistry to return to its normal state if it is reacting to pain signals.
There are many ways to cope with post-TBI depression. Talking to close family or friends about how you are feeling is a great way to minimize isolation. Isolation exacerbates depression so it should be avoided if at all possible. If you are struggling with being in a crowd or around people, try to meet with one close friend in a quiet location. Research shows that listening to uplifting music, engaging in a creative activity, or reading a book are effective ways of coping with depression. Exercise is also a strongly proven antidote for depression. Just a few minutes each day will produce significant results. Most importantly, seek the support of a professional who has experience treating patients with TBI.
A TBI patient’s prognosis as it relates to depression is highly dependent on the type of TBI they experience. Patients who sustain an mTBI (minor TBI) tend to recover quickly, within one to three weeks, with little to no lingering symptoms. In these cases, depression is less of a concern. In a small percentage of mTBI injuries, patients may experience side effects for up to three months, and as mentioned previously, longer recovery times can lead to long-term depression.
Complicated mTBIs can cause more cognitive impairment than uncomplicated mTBIs, and in the latter, recovery can take up to six months and mirror the recuperation associated with a moderate TBI.
Sustaining a TBI can be a lifelong challenge and can require a range of resources and strategies that extend far beyond initial recovery. When depression is a factor in recovery, it can be even more challenging. Neuropsychologists and neuropsychiatrists are not only important for diagnosis and first phases of treatment but can also be an important part of a long-term treatment plan which addresses mood and emotional wellbeing, helping TBI survivors thrive in their daily lives long after their injury.
Traumatic brain injury (tbi) is a significant health concern worldwide, affecting millions of people each year. It occurs when an external force, such as a blow or jolt to the head, causes damage to the brain. Symptoms may range from mild concussions to severe injuries leading to permanent disability or death.
Most treatment options focus primarily on managing symptoms and preventing further damage, with limited success in reversing the injury. However, recent advancements in regenerative medicine, particularly stem cell therapy, have generated excitement in the medical community and among patients for their potential to heal the brain and restore lost cognitive functions.
Stem cell therapy could promote brain repair, offering hope to individuals living with the long-term effects of TBI. This article will explore how stem cell therapy works and the promise it holds for treating TBI.
THE SCIENCE BEHIND STEM CELL THERAPY
Stem cell therapy involves using undifferentiated cells — cells that have the ability to become specialized into various types of tissues and organs — to repair or replace damaged cells in the body. These cells have key properties that make them highly promising for TBI treatment.
For example, stem cells have the ability to differentiate into various types of specialized cells, including neurons, which are the cells affected after a TBI. This regenerative potential is what makes stem cells a compelling option for healing brain tissue. In addition to their regenerative capacity, stem cells can reduce inflammation and provide protection to existing neurons. This is particularly important in TBI, as inflammation in the brain is a key contributor to secondary damage following injury.
HOW STEM CELL THERAPY IS REVOLUTIONIZING TBI TREATMENT
The potential of stem cell therapy to treat TBI lies in its ability to promote brain repair and recovery. Here’s how stem cell therapy works in the context of TBI:
Neurogenesis (Regeneration of Neurons): After a TBI, neurons (brain cells) may be damaged or die, leading to cognitive and motor impairments. Stem cells have the potential to replace lost neurons by differentiating into functional brain cells. This can improve brain function and potentially restore lost abilities such as memory, motor skills and speech.
Reduction of Inflammation: In the aftermath of a brain injury, inflammation often exacerbates damage. Stem cells can modulate the immune response, reducing inflammation and preventing further harm to healthy brain tissue. This could be particularly valuable in the days and weeks following an injury.
Improvement of Blood Flow to the Brain: Stem cells can also help restore damaged blood vessels, improving circulation in the brain. Better blood flow can support the healing process by delivering essential nutrients and oxygen to injured areas.
Functional Recovery: In addition to directly repairing brain tissue, stem cells can promote the growth of new connections between surviving neurons. This network remodelling could help restore lost functions and improve the overall health of the brain.
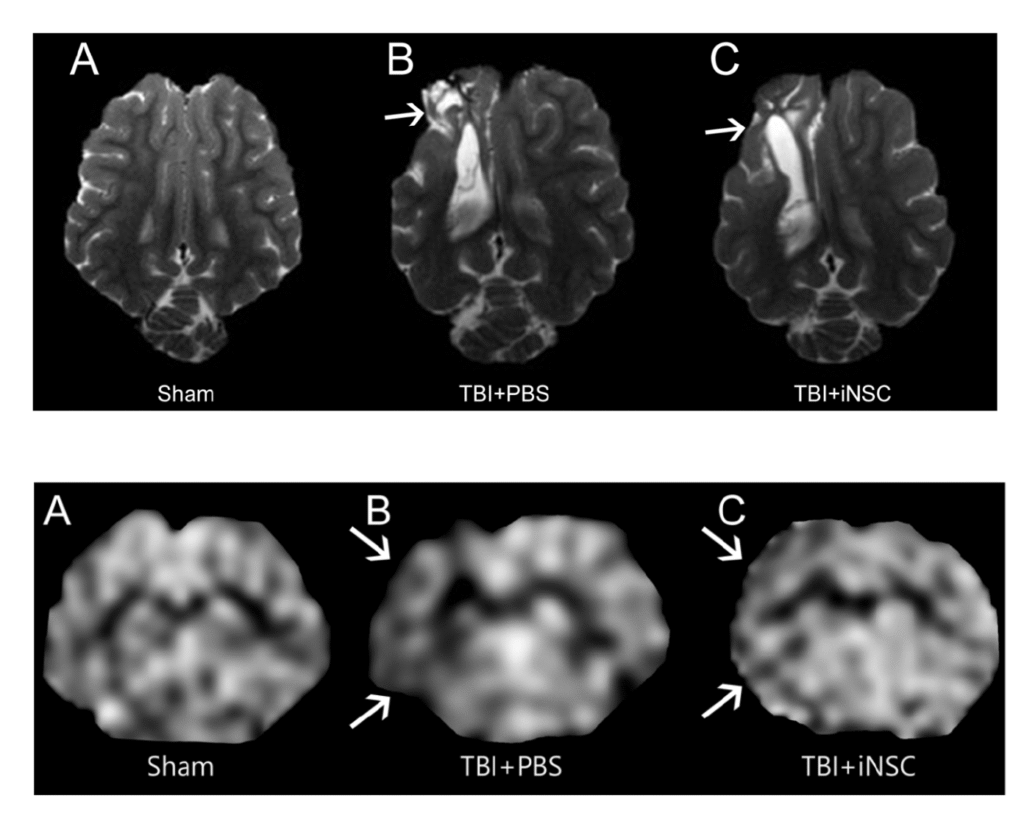
Top: Transplantation of stem cells decreased tissue atrophy and lesion volume in a study using piglet brains effected by TBI. Bottom: Transplantation of stem cells preserved cerebral blood flow in a study using piglet brains effected by TBI.
Scans courtesy of: Schantz, S. L., Sneed, S. E., Fagan, M. M., Golan, M. E., Cheek, S. R., Kinder, H. A., Duberstein, K. J., Kaiser, E. E., & West, F. D. (2024). Human-Induced Pluripotent Stem Cell-Derived Neural Stem Cell Therapy Limits Tissue Damage and Promotes Tissue Regeneration and Functional Recovery in a Pediatric Piglet Traumatic-Brain-Injury Model. Biomedicines, 12(8), 1663. https://doi.org/10.3390/biomedicines12081663
THE ROAD AHEAD: A BRIGHT FUTURE FOR TBI PATIENTS
The advances in stem cell therapy in recent years suggest a bright future for this innovative treatment. Stem cell-based therapies are set to become a standard part of treatment for TBI. With continued investment in research and a growing understanding of the brain’s regenerative capabilities, stem cell therapy holds immense promise for TBI patients.
IN CONCLUSION
Stem cell therapy represents one of the most exciting innovations in medicine, particularly in the treatment of traumatic brain injury. It has the potential to offer not just symptom management, but actual recovery and healing of the brain. The promise of stem cell therapy to treat TBI could transform the lives of millions of people who suffer from brain injuries, restoring their abilities and improving their quality of life. With continued innovation and collaboration in the scientific community, stem cell therapy is becoming a breakthrough treatment that is revolutionizing the field of TBI recovery.
Contribute to the TBI Times





